
This Article From Issue
May-June 2020
Volume 108, Number 3
Page 138
Whether you know it or not, odds are a spider is within reach.
Wherever you are.
Like, right now.
Lucky you! These lovable weirdos do lots of excellent work all around us (thanks for the free pest control). But outside regular working hours, some species have odd personal lives. Here are five strange habits of some of the most common spider species creeping around homes and lawns across much of North America.
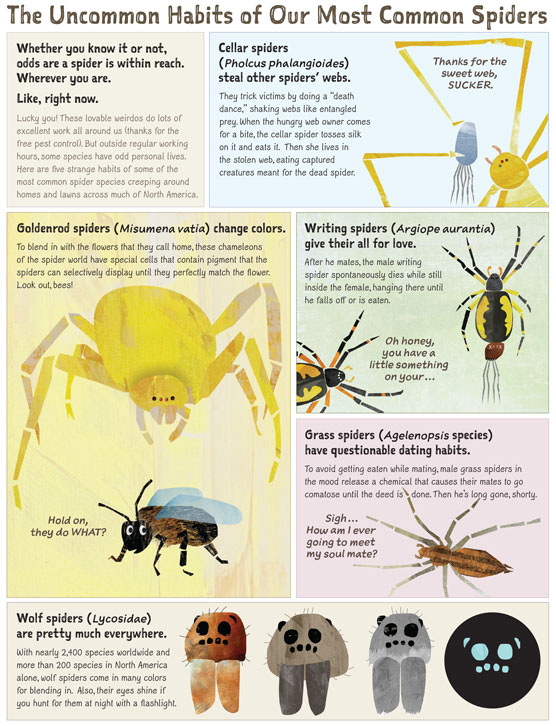
Infographic by Eleanor Spicer Rice and Neil McCoy.
Cellar spiders (Pholcus phalangioides) steal other spiders' webs.
They trick victims by doing a "death dance," shaking webs like entangled prey. When the hungry web owner comes for a bite, the cellar spider tosses silk on it and eats it. Then she lives in the stolen web, eating captured creatures meant for the dead spider.
Goldenrod spiders (Misumena vatia) change colors.
To blend in with the flowers that they call home, these chameleons of the spider world have special cells that contain pigment that the spiders can selectively display until they perfectly match the flower. Look out, bees!
Writing spiders (Argiope aurantia) give their all for love.
After he mates, the male writing spider spontaneously dies while still inside the female, hanging there until he falls off or is eaten.
Grass spiders (Agelenopsis species) have questionable dating habits.
To avoid getting eaten while mating, male grass spiders in the mood release a chemical that causes their mates to go comatose until the deed is done. Then he's long gone, shorty.
Wolf spiders (Lycosidae) are pretty much everywhere.
With nearly 2,400 species worldwide and more than 200 species in North America alone, wolf spiders come in many colors for blending in. Also, their eyes shine if you hunt for them at night with a flashlight.
American Scientist Comments and Discussion
To discuss our articles or comment on them, please share them and tag American Scientist on social media platforms. Here are links to our profiles on Twitter, Facebook, and LinkedIn.
If we re-share your post, we will moderate comments/discussion following our comments policy.