Curious Chemistry Guides Hydrangea Colors
By Henry Schreiber
The blooms’ varied hues trace back to both soil pH and the right additives. Could different combinations produce new tints never seen in nature?
The blooms’ varied hues trace back to both soil pH and the right additives. Could different combinations produce new tints never seen in nature?

DOI: 10.1511/2014.111.444
One of the world’s most popular ornamental flowers conceals a bouquet of biological and biochemical surprises. The iconic “snowball” shaped blooms of Hydrangea macrophylla (big-leafed hydrangea) are a common staple of backyard gardens.
Many other, closely related hydrangea species are likewise known for their bountiful, showy, long-lasting blossoms, making them popular for both landscaping and the cut flower market. And their popularity continues to grow: Every year, gardening catalogs add new cultivars of these attractive plants. Hydrangeas are ubiquitous—but they are not what they seem.

Wikimedia Commons
For starters, the bloom of the hydrangea is not a true flower, but an inflorescence: Sepals, or modified leaves, make up most of the bloom and overshadow the small, almost unnoticeable fertile floral portions at the center.
The bloom colors are what really make the hydrangea stand out: They range from pink to blue, including all shades of lavender to violet to purple, as well as green and white. Color intensities run the gamut from vibrant to pastel. Noticeably absent from the kaleidoscope of possible hydrangea colors are yellows and oranges.
Hydrangea colors are not what they seem, either; they are not the result of a variety of different pigments, as is the case for flowers such as roses or tulips. They are more akin to the colors seen in litmus paper, the chemically treated strips classically used to determine whether solutions are acidic or basic. At the molecular level, acids are proton (or hydrogen ion) donators and bases are proton acceptors in chemical reactions. When one dips blue litmus paper into an acidic solution (pH < 7, where pH is a measure of the concentration of hydrogen ions), the paper turns red, whereas red litmus paper changes to blue in the presence of a basic solution (pH > 7).
In a similar fashion, the color of many hydrangea blooms acts as a natural pH indicator for the soil in which the plant grows. Such blooms have blue sepals when the shrub grows in acidic soil, but develop red or pink sepals when grown in neutral to basic soils. The hydrangea’s bloom color reveals the pH of the soil, but with its distinguishing colors being the reverse of those for litmus paper. The hydrangea is unique among plants in this ability to indicate soil acidity.
Because of this trait, gardeners can chemically manipulate hydrangea bloom colors using soil additives. In fact, a hydrangea can have different bloom colors on the same bush if the roots of the plant sample soils of differing pH. Homespun recipes abound for changing the pink blooms of a hydrangea to blue: pouring vinegar or lemon juice on the soil; mulching the plant with coffee grounds, citrus fruit rinds, or pine tree needles; or burying rusty nails, old tin cans, or copper pennies next to the bush. All these strategies tend to turn soil more acidic, and eventually transform the bloom color to blue.
Hydrangea colors turn out to be even more complicated that that, however. Soil acidity actually is not the underlying chemical mechanism behind the color change. The answer goes even deeper into the connection between soil composition and sepal color— a connection that has inspired our ongoing research into the biochemistry of these flowering plants.
Hydrangea colors ultimately depend on the availability of aluminum ions (Al3+) within the soil. The role of aluminum has been known since the 1940s, but it did not reach the mainstream horticultural literature until about the past two decades, and the exact mechanism was only recently defined. Aluminum ions are mobile in acidic soil because of the ready availability of other ions they can react with, which can be taken up into the hydrangea to the bloom where they interact with the normally red pigment. But in neutral to basic soil, the ions combine with hydroxide ions (OH-) to form immobile aluminum hydroxide, Al(OH)3. Consequently, for the bluing of hydrangea blooms, one needs both aluminum ions and acidic soil. The best soil additive for bluing is one that contributes both, such as commercially available aluminum sulfate, Al2(SO4)3. Conversely, if one wishes to change blue-blooming hydrangea to red-blooming, adding lime (calcium hydroxide, Ca(OH)2) results in basic soil and the desired color transition.
However, such imposed red-to-blue or blue-to-red color changes do not happen instantaneously; it often takes one or two growing seasons to instill the desired color on shrubs within one’s flower gardens. Growers of hydrangeas with blue blooms must regularly water with aluminum sulfate drenches onto the potted medium to maintain the needed levels to force the desired blue coloration (but they cannot water too often or the excess Al3+ will kill the plant).
The chemistry of aluminum in soil establishes its different properties under acidic and basic conditions. In acidic soils, aluminum occurs in what are called coordination complexes, with Al3+ ions at the center, surrounded by bonded strings of other molecules. These aluminum ions can travel from soil into the plant. But at neutral to basic pH, aluminum precipitates as aluminum hydroxide, making it unavailable for incorporation into the shrub. Lavenders, magentas, violets, and purples appear as bloom colors in transitional soil pHs, with aluminum ions only somewhat available to the hydrangea roots.

Photograph courtesy of the author.
At extremely high pHs or very basic conditions, such as in hydroponic systems where plants are grown in nutrient water without soil, aluminum ions such as Al(OH)4-, called the tetrahydroxyaluminate ion, become stable, so they no longer precipitate and are available as aluminum ions once again to the hydrangea. Indeed, at these extremely high pHs, the hydrangea bloom becomes blue, shortly before the plant dies from the extreme basicity, which causes cellular damage. On the other hand, because aluminum phosphate has limited solubility, it is also possible to block aluminum ion availability, even in acidic soils, through the use of high-phosphate fertilizers.
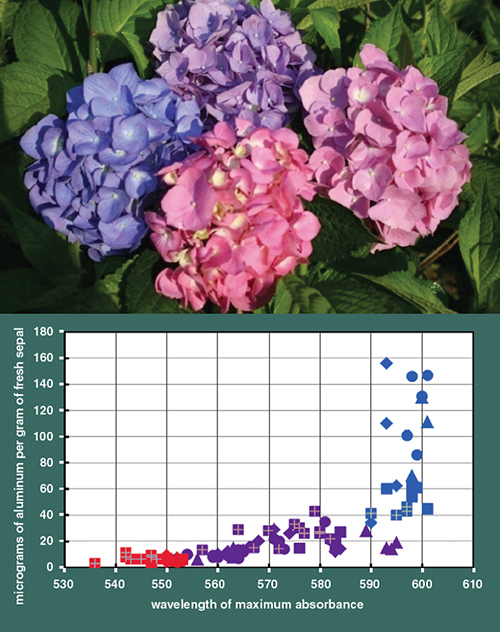
Illustration by Tom Dunne. Photograph courtesy of the author.
Data on the sepal’s aluminum content (see figure above) show that red sepals possess essentially no aluminum. But a little aluminum goes a long way toward bluing the bloom. At a threshold of only about 40 micrograms of aluminum per gram of fresh sepal, hydrangea sepals turn blue, but they don’t become bluer with yet more aluminum. Intermediate sepal colors of lavenders to purples have aluminum contents lower than this threshold.
Thus, it’s all about the availability of aluminum ions in the soil for the generation of the blue sepal color in hydrangea blooms, with the soil pH just being a necessary facilitator of this aluminum mobility and availability.
In other cases where a plant has a flower that can be different colors, it is usually because the underlying pigments are likewise different, or the proportion of its pigments changes. However, the hydrangea is additionally unique because the color comes from only a sole pigment, delphinidin-3-glucoside (which is in the anthocyanin family, the same group that turns leaves red in autumn and gives berries their color). The underlying chemical system is thus, in a sense, relatively simple.
The color of the delphinidin-3- glucoside, as well as other anthocyanins, is a function of its molecular structure, which determines what wavelengths of light it absorbs. These molecules consist of a central three-ring carbon chain with one oxygen substitution, called a flavylium cation at low pH, to which various sugars are connected. The anthocyanin loses one or more hydrogen ions as the pH environment changes, which alters its absorbance spectra.
What goes on at the pigment level inside the cell is actually further proof that the soil pH is not directly responsible for the color switch, but rather mostly an indicator of aluminum ion availability. The internal cell pH remains constant for both red and blue sepals. The flavylium cation is red and stable at low pH, the opposite of the overall bloom color under acidic conditions. But under neutral conditions it transforms to the purple form of what is called a quinoidal base, meaning the molecule has lost a hydrogen ion and rearranged its double bonds. At basic pHs, the quinoidal base anion forms with a blue structure upon loss of another hydrogen ion and further rearrangement of the double bonds in the core delphinidin component of the pigment.
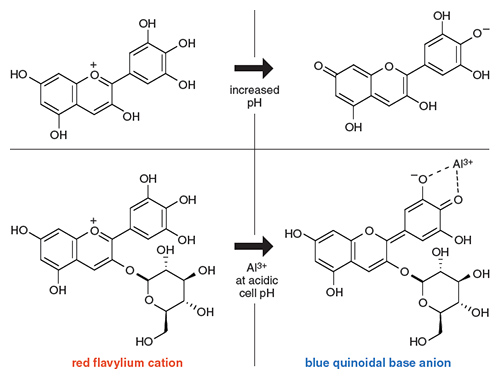
Illustration by Tom Dunne.
On the other hand, studies have shown that there is a way to stabilize this blue quinoidal base anion in an acidic cell medium. Aluminum ions will complex with the normally red pigment, as also shown in the figure above, for delphindin-3-glucoside, and result in additional bluing. Once again, the presence of Al3+ becomes the key for the bluing of hydrangea sepals—both at the molecular level and in the field. Its presence circumvents the need for a high pH inside the cells to create the blue structure.
To ascertain the exact nature of the Al3+-anthocyanin complex, my research group undertook chemical modeling studies using acidic ethanol as a solvent. (Anthocyanins react with water to form yellowish to colorless structures called chalcones, which don’t behave chemically the same way as the pigments, so water can’t be used readily as a solvent.) We added aluminum chloride, which will break apart under the acidic conditions into aluminum ions, to a constant concentration of delphinidin, or delphinidin-3-glucoside. (The sugar substituent on the core delphinidin did not affect color appreciably. We also repeated this experiment with a direct extract from hydrangeas, with analogous results.)
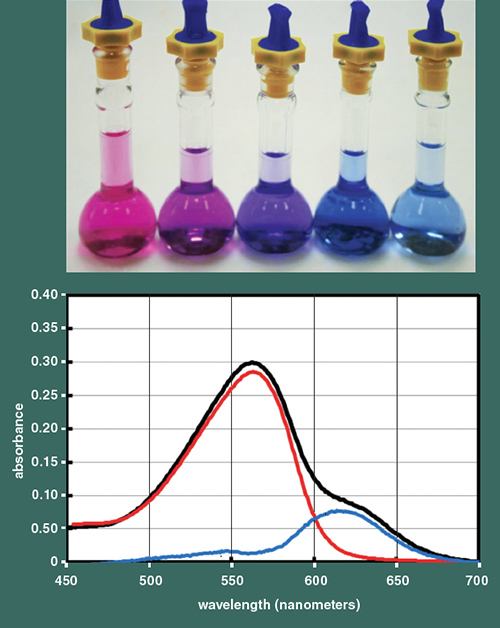
Photograph courtesy of the author. Illustration by Tom Dunne.
The figure above illustrates a series of samples in which ever-increasing amounts of Al3+ are added to delphinidin in the solvent. The color systematically changes from red to blue, through varying shades of purple, with the increasing Al3+. Once blue, the intensity of the blue plateaus much like in the natural system; the color does not become any bluer with even more Al3+. We used a type of spectroscopy, which in this case excites the molecules with a high-energy visible light, so they absorb a wavelength characteristic of their structure. These data let us resolve the mechanism by which the Al3+ complexed with the delphinidin. A peak at a wavelength of about 620 nanometers is characteristic of the blue quinoidal base anion, the structure complexed with the aluminum. As the amount of Al3+ increases, the intensity of this peak (or the amount of the complex) increases, but finally plateaus.
The second peak we found, at a lower wavelength, is characteristic of the flavylium cation. As the amount of Al3+ increases, its intensity tends to decrease, but the position of the peak steadily shifts to higher wavelengths until it too reaches a constant value—that is, the original red color of the flavylium cation transitions to a blue color. Thus, we discovered two contributions to the bluing of the solution: the previously acknowledged formation of the blue quinoidal base anion complexed with the Al3+, and the steady transition of the red to the blue flavylium cation.
To develop a complete picture of the Al3+-delphinidin complex, we pondered why the flavylium cation also went through a color transition. We collected other pertinent evidence showing that only about half of the available delphinidin molecules would form complexes (and create the blue quinoidal base anion structures) with the Al3+, regardless of how much of the latter was added. Evidently, each mechanism produces half of the final blue product. This behavior is often characteristic of stacking, when two molecules sit one on top of another like two slices of bread, but it turns out what’s happening is more complex than a simple stack.
The Al3+-delphinidin’s quinoidal base anion constitutes the primary blue complex. A second part of the complex, leading to enhanced bluing, is the stacking of a flavylium cation on this primary complex. The flavylium cation and quinoidal base anion are kept together not only by the electrostatic attraction that results from their opposite charges, but also, because their cyclic structures are similar, the molecules’ electron orbitals can align themselves for further stabilization. Thus, we have generated both a chemical mechanism and a model for the bluing.
Note that the Al3+ serves as an anchor for this complex, probably attached to a phosphate network within the sepals’ cells, and not as a central ion for the complex. Indeed, we are finding that the aluminum ion is not material in the color generation, only for its stabilization, so that replacement of this metal by other metal complexing agents should not change the color. Our tests have shown that scandium (Sc3+, a common surrogate for Al3+), gallium (Ga3+, in the same periodic family as Al3+), tin, molybdenum, uranium, and other metal ions behaved analogously with delphinidin and formed blue complexes, albeit not as effectively as Al3+. That is, the chemical mechanism for bluing was the same, but the effectiveness of the specific metal ions in creating the resulting stacked complex was not.
Others have further characterized the Al3+-delphinidin complex and have shown the stacked flavylium cation is skewed at an angle from the underlying quinoidal base anion. The naturally forming complex inside the cellular environment of the hydrangea sepals has additional stacking and stabilizing with other co-pigments in the system. Such co-pigments, likely a unique mix of which exist in each cultivar, are a bit misnamed because they only serve to help stabilize the blue complex and do not contribute to the color. But the result is a complex probably in the shape of a helical spiral, rather than a simple stack.
A key step in the bluing of hydrangea sepals relies on getting Al3+ into the plant and transporting it to the sepals, but as seems to be a theme with hydrangeas, it turns out that there’s another step in the process of aluminum transport. Al3+ is mobile under acidic soil conditions and, in response to its stimulus, the roots of the hydrangea exude citric acid (C6H8O7). Consequently, a solution of citrate ions (C6H5O73-) and citric acid forms around the roots at relative concentrations that are specific to the soil pH. Al3+ then establishes a stable complex with the citrate ions, which is absorbable into the roots of the hydrangea. The plant transports Al3+ throughout as this citrate complex. Other Al3+-tolerant plants, such as buckwheat and rye, likewise exude simple organic acids to detoxify aluminum. In fact, such strategies are becoming quite important in cultivating crops that are being both bred and genetically engineered to survive in acidic Al3+-rich soils.
This citrate complex is crucial for not only the incorporation of Al3+ into the roots but also the constant circulation of Al3+ throughout the plant, as shown in the figure at right. The hydrangea sepals actually do not concentrate the Al3+, as all leaves on the hydrangea possess about the same concentration of Al3+ as the sepals (but only the sepals have the correct pigments to react with the ions). Because sepals are simply modified leaves, such behavior might be expected.

Photographs courtesy of the author.
In fact, it is possible to change a hydrangea’s color without affecting the soil chemistry at all. We have been able to circumvent the need for Al3+ assimilation through the roots and subsequently transport to the sepals by developing a spray that introduces Al3+ directly into the sepals. By dissolving appropriate amounts of Al3+ in a buffered citrate–citric acid solution, we have changed red sepals to blue in a period of several days, as shown in the figure at above right. This outcome is further evidence that the bluing depends on the presence of Al3+ in the sepals. The reverse spray to change blue sepals back to red has proven to be a more challenging chemical problem. That is, the Al3+-delphinidin complex, once formed, is difficult to break apart inside the plant.

Illustration by Tom Dunne.
The illustration above provides a comprehensive model for the chemical incorporation of aluminum ions into the hydrangea, resulting in the bluing of sepals. The ease of bluing in hydrangea sepals depends on the relative concentrations of the delphinidin-3-glucoside and Al3+, as the number of Al3+ molecules has to be in excess of about 3 to 10 times the number of pigment molecules. Thus, the less intense the coloration, the less the pigment concentration, and the less Al3+ needed to attain the molecular excess. In addition, not all hydrangeas are equal in their ability to exude citric acid and incorporate Al3+ into the plant.
Watch the chemistry behind hydrangea colors in action:
The biochemistry of hydrangeas opens up the possibility of developing new colors that are both interesting scientific experiments and potentially beautiful additions to the garden. The genetics of a particular hydrangea cultivar controls the amount of delphinidin-3- glucoside in the sepals. This content varies from zero for white cultivars to greater than 700 micrograms of delphinidin-3- glucoside per gram of fresh sepal in the most intensely colored breeds, as shown in the figure below. The concentration of pigment indicates the intensity of the coloration, from pastel to vivid, not whether the color is red or blue.

Photograph courtesy of the author. Illustration by Tom Dunne.
Hydrangea cultivars that bloom on old wood, or the previous year’s growth, are susceptible to the loss of their blooms for an entire growing season upon a cold winter or a late frost, as the buds from the previous year freeze. Several new cultivars, most notably the popular “Endless Summer” cultivars, set their blooms on new wood, the current year’s growth, and will flower each year, unaffected by the weather of the winter or spring. However, such cultivars usually have more subdued colors than other hydrangea cultivars, for reasons yet unclear. One direction for breeding and genetic engineering has been to generate similar cultivars with brighter or more intense coloration.
Incorporating new natural pigments could yield hydrangea blooms that fluoresce at night as well.
Alternatively, we tried a chemical approach to enhance the color brightness by infusing magnesium ions (Mg2+) into the plant, because other researchers have shown that such an approach brightened the color of grapes as well as other floral anthocyanin-based blooms, although the mechanism remains unknown. Surprisingly, instead of intensifying the coloration of the hydrangea sepals, we were able to produce sepals with red, white, and blue colors simultaneously (see figure below) on certain cultivars. It’s still an open research question as to why and how this happens.

Photographs courtesy of the author.
But these results inspired us to see if it is possible to manipulate hydrangea chemistry to produce totally new colors. Hydrangeas lack the necessary pigment to generate yellow or orange sepals. Our initial strategy to produce these colors was to replace Al3+ with some other metal, at first using solutions in the laboratory. However, the chemical mechanism we found for bluing indicated that such an approach might not work, because aluminum did not really contribute to the blue color, but only acted as an anchor for the blue form of the delphinidin core. Therefore, we were not surprised when our experiments with other metals that complexed with the delphinidin generated only subtle differences in blue hues.
But in another case of serendipity, during attempts to create a slightly different shade of blue with the infusion of the molybdate ion (MoO42-) through the cut stems of red blooms, the sepals did not turn blue like the solutions, but showed yellowing. The yellowing occurred via a different mechanism than the bluing, observed not on the top surface but on the lighter bottom surface of the sepals. Accordingly, we changed strategy and started over with a white bloom of the hydrangea to generate, with success, the yellow color. We hypothesize that the normally colorless MoO42- bonds with the same phosphate groups that usually anchor Al3+, creating a yellow phospho- molybdate entity that gets stuck on the bottom surface of the sepals, for reasons we are still trying to determine.
Similar to some hydrangea cultivars being better “bluers” than others, it appears that a sepal’s yellowing ability is also a function of the cultivar. Such yellowing, as shown in the figure above, has only been successful thus far through cut stem infusions and sprayings. Introduction of the molybdate ion into the hydrangea shrub through the soil has proven toxic to the plant, but long-term experiments are in progress exposing hydrangeas to very low concentrations of MoO42- in the soil to create a viable plant with attractive, strongly yellow-colored sepals.
Another result of our research is the production of unique red-blue patterns on hydrangea sepals by controlled aluminum diffusion through cut stems and by direct sprayings onto blooms. Whereas the incorporation of Al3+ through the roots always resulted in the relatively homogeneous distribution of blue color in the sepals, the rapid forcing of aluminum into the initially red sepals quickly generates unusual and novel patterns of red and blue.
There is still much to learn about natural changes in the color in hydrangea blooms. Near the end of the growing season, the sepals of the blooms of some plants flip over and change colors from blue to red, even though they retain the same aluminum content. The reason for this transformation is still unknown. A possible explanation is that the operational anthocyanin changes its composition, perhaps from the delphinidin- 3-glucoside to cyanidin-3-glucoside, which requires much more of an aluminum excess to stabilize the blue complex.
One last example of our current research on hydrangea coloration focuses on the blooms and leaves of the oak-leaf hydrangea, H. quercifolia. The green leaves of this hydrangea turn red in the autumn, much like its blooms age from white to red, with increasing intensity over time. Such blooms and leaves do not turn blue in the presence of Al3+ as for H. macrophylla sepals, probably once again due to differences in anthocyanin types and levels. But we hope with more research, we may be able to generate intensely blue, “Smurf”-colored oak-leaf hydrangea leaves during the fall months. We envision beds of blue-leafed hydrangeas to complement the autumn spectrum of reds and golds of other shrubs and trees.
In the future, we plan to investigate the incorporation of entirely new natural pigments, not just the insertion of other metals, from various flowers to the hydrangea sepals. Perhaps we can attain this result via a spray to localize the desired changes—such that the changes are permanent for selective blooms without affecting neighboring plants and blooms. We imagine not only color changes but also controlling other properties, such as fluorescence, so that one can achieve hydrangea blooms that glow at night as well. For example, we are investigating the transfer of the fluorescent betaxanthin pigment from yellow flowers of the night-blooming four o’clocks (Mirabilis jalapa) and of portulacas to hydrangea blooms. Adding specific metal ions, such as those of rare earth elements, to the betaxanthin pigment seems to fine-tune the desired fluorescent hue.
It seems that when it comes to hydrangea colors, there are always more chemical mysteries awaiting a solution. And exploration of the coloring of hydrangea blooms illustrates that metal ions as well as pH play a key role in being able to create designer colors for flowers. Although such chemical manipulation of these colors has been underused in the past, this method may represent a fertile approach to generate novel floral colors for the future.
Click "American Scientist" to access home page
American Scientist Comments and Discussion
To discuss our articles or comment on them, please share them and tag American Scientist on social media platforms. Here are links to our profiles on Twitter, Facebook, and LinkedIn.
If we re-share your post, we will moderate comments/discussion following our comments policy.