Health and Human Society
By Clyde Hertzman
Wealthier nations are not always healthier, and efforts to improve health can be swamped by the effects of inequality and conflict
Wealthier nations are not always healthier, and efforts to improve health can be swamped by the effects of inequality and conflict

DOI: 10.1511/2001.40.538
Two years ago I visited the Gilgit region of Northern Pakistan to learn about the revolution in health that had taken place there over the preceding 15 years. With assistance from the Aga Khan Foundation, a series of new institutions were developed in the region: a women's organization for agricultural diversification, economic participation and mutual aid; schooling for girls; a rural sanitation service; and a system of primary health care. The impact of the changes was breathtaking.
During this short period the infant mortality rate fell more than four-fifths to approach Western levels. Birth rates were cut in half. The main concerns of village women had become the familiar modern problems of balancing work and home life, since both male and female children were now at school and not available to mind younger siblings.
The Gilgit region had recapitulated, in 15 short years, a trajectory of health development that took Western societies two centuries to complete. In their transformation the people of Gilgit made their region a case study in the "determinants of health"—the ways in which the prospects for health and well-being in human societies are embedded in the environments where people grow up, live and learn. These underlying conditions are complemented but not preempted by access to medical services.

K. L. Kamat
Just as some people are healthier than others, it is apparent that some societies are healthier than others, and not simply because they can afford good nutrition and advanced medical care. Research into the determinants of health has accelerated in the past two decades, going beyond the study of risk factors for individual diseases to get at the underlying forces that explain larger patterns of disparities in health status. The sources of insight here are diverse: international comparisons; birth, working-age and elderly cohorts; ethnographies; primate models of health and society; and biological studies of social influences on the development and function of the brain and "host defense" systems. Through this research we have come to understand how the health of a population as a whole is influenced by social and economic conditions and the "psychosocial" environment on the one hand, and by experiences during sensitive periods in early biological development, on the other. We understand why the people of Gilgit are now healthier.
In general terms, the emerging story of the determinants of health goes like this: The history, wealth, and social and institutional arrangements of human societies powerfully influence the material and psychosocial environments (and life chances) experienced by their members. The developing individual, from the time of earliest biological promise or vulnerability onward, experiences these conditions as a moment-to-moment, day-to-day, year-to-year interaction with the intimate, civic, and broader socioeconomic and physical environment.
To a certain extent, these environments shape human biology and set the prospects for health (as well as well-being and competence) throughout an individual's life course. This manifests first through a latent effect, with early development (encompassing factors such as genetic endowment, infant nutrition and nurturance) affecting health later in life, irrespective of intervening experiences. Other research has shown a pathway effect, wherein health is, in part, determined by living conditions that are associated with well-traveled socioeconomic careers, and a cumulative effect: Personal resources or resiliencies, as well as injuries or vulnerabilities, can accumulate through youth, maturity and old age.
These influences are expressed through a phenomenon known as the socioeconomic gradient in health. The average health status of members within every society on earth increases in a stepwise fashion as one ascends from the bottom of the social ladder (defined, variously, by income, education or occupation) to the top. During the 1980s and early 1990s, a series of investigations demonstrated that the gradient could not be explained by reverse causality or health selection. Now, those working at the frontier of population health aim to understand, in greater detail, how societal processes generate the gradient and why it has a steeper slope in some societies than in others.
So far, this research has generated a series of robust, yet discrete, observations that can be connected, just as children connect the dots to make a picture, to produce the outline that follows. In this case, what results when you connect the dots is an understanding of the determinants of health in human societies.
Annette deFerrari
1. Increased life expectancy in wealthy societies is not primarily due to the provision of individual health care services.
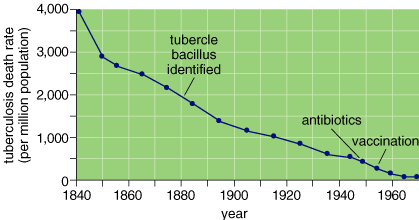
In exhaustive research published during the 1970s, Thomas McKeown, a British epidemiologist, demonstrated conclusively that clinically effective vaccinations and antibiotic treatments did not deserve credit for the fact that the major infectious diseases of antiquity, such as tuberculosis, had precipitously declined in his country during the previous century and a half. These diseases had been the major causes of mortality for centuries, yet effective clinical prevention and treatment interventions were developed and implemented after the death rates from these conditions had declined by approximately 90 per cent. Thus, in direct contradiction to common belief, the vaccine and antibiotic revolution played a relatively minor role in lengthening human life expectancies in the modern world's wealthy societies.
The principal factors responsible for increasing life expectancy from less than 50 years to more than 70 years in wealthy countries are to be found outside the health care system as it is traditionally defined and, instead, in the broader socioeconomic environment. Socioeconomic factors that could be related to improved health include improvements in housing, water supply, pollution control, nutrition, child spacing, working conditions, education and a wide range of psychosocial conditions that are thought to be most hospitable in prosperous, tolerant, democratic societies with strong civic communities. Some of these conditions have improved through a conscious effort to improve the health and well-being of the population. But others have been by-products of societal development per se and not acts of conscious human agency. The question currently being debated here is, "how important has conscious human agency been in translating socioeconomic progress into health?"
2. Historically and internationally, increasing wealth has been a strong correlate of improved health status. This is much less the case among the world's wealthiest countries today.
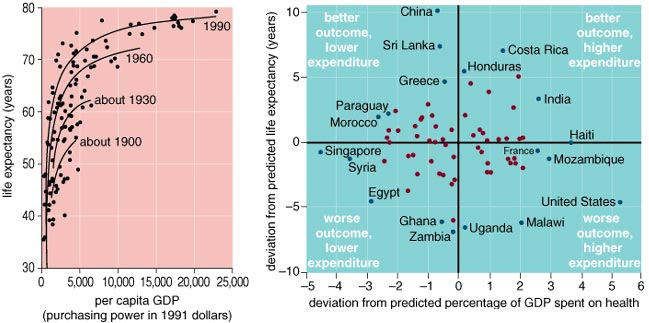
There are substantial differences in health status among wealthy countries today. The average life-expectancy difference between the "healthiest" of the world's wealthy countries and the least healthy of this group is approximately five years. This five-year life-expectancy difference (between 74 and 79 years) is generated by an approximate doubling of age-specific mortality during the early adult years (ages 25–64). So the differences in health status among the wealthy countries of the world are actually quite substantial. A country does not leap from the low end of this range to the high end simply by investing in medicine: The evidence is conclusive that national differences in spending on health-care services do not explain these differences. Scatter plots of health-care spending versus life expectancy among wealthy countries consistently show no association whatsoever.
Annette deFerrari
Differences in wealth (per capita Gross Domestic Product, or GDP) among the world's wealthiest countries are only weakly associated with health-status differences among them. In the early part of the century there was a strong correlation between increasing GDP and increased life expectancy for all countries in the world. As the century progressed and wealthy countries became wealthier, a "flat of the curve" gradually developed where this association weakened considerably. By 1990 the flat of the curve included those countries with GDPs per capita greater than $11,000. Because of this pattern, it has been argued that relative income and social status are more important determinants of health than absolute income in wealthy countries, operating through a process of "psychosocial comparisons" that are, predictably, more invidious and health-damaging in less equal, more hierarchical societies.
3. Differences in health status among wealthy societies, and among regions within them, are a function of differences in the character of the socioeconomic environment and civil society, and in societal investment in the early parts of the life course.
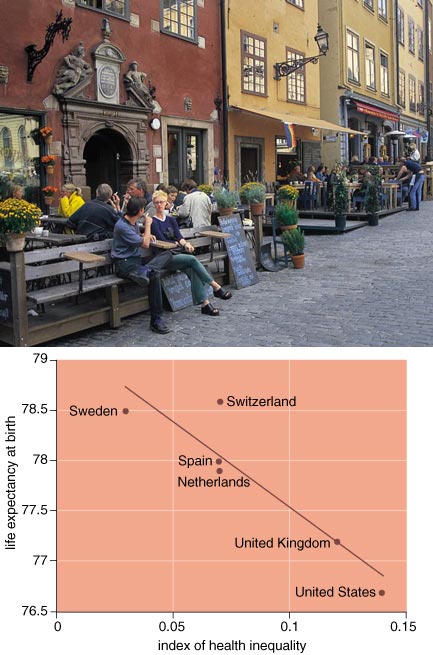
Recently, a team of Canadians and Americans collaborated on a comparison of mortality and income inequality among the 10 Canadian provinces and the 50 U.S. states. Although this is only one of many international comparisons of income distribution and health, it is the most important one because it best overcame the methodological problems associated with comparing income distributions in different societies.
Overall, higher mortality was closely associated with higher levels of income inequality across the 60 provinces and states. The range of income inequality among the states was found to be very large, whereas among the Canadian provinces the range was much narrower. Compared to the U.S. states, all the Canadian provinces had a relatively egalitarian income distribution. Accordingly, all Canadian provinces fell at the lowest level of mortality among the entities studied, whereas the states with the highest level of income inequality had mortality rates, after adjustment for age, which were 30 to 60 percent higher than those of the Canadian provinces. This pattern could not be explained away by "pockets of poverty" effects or any other competing explanatory model.
Historically, those regions of the United States with the most unequal income distributions tend to be those that began as vertically organized, plantation economies, whereas those with more equal income distributions began as horizontal, freeholder economies. The former group tends to have higher levels of social distrust, weaker social safety nets, less investment in education and higher levels of gender inequality.
4. There is a complex interplay between factors associated with the psychosocial environment and the material living conditions that act to support or undermine health status. These determinants of health act at various levels of societal aggregation.
From the time of conception and throughout the life course, population health is determined at three levels of societal aggregation. At the broadest level is the national socioeconomic environment: the wealth of a society and how it is distributed. At the next, more immediate level is civil society. To what extent do the institutions of civil society, as they are encountered on a daily basis, buffer or exacerbate the stresses of daily living, and promote or undermine living conditions that, over the long-term, are compatible with health and well-being? This question encompasses a range of factors: social trust, participation, psychosocial working conditions, the responsiveness of institutions to individual needs, and neighborhood cohesion and safety, to name a few. Finally, at the most intimate level, relations in the private realm are fundamental—that is, the quality of stimulation, support and nurturance that individuals experience, especially in the earliest stages of life.

Annette deFerrari
In order to understand what makes some societies healthier than others, it is necessary to consider these various levels of social aggregation simultaneously. In introducing their recent edited volume Is Inequality Bad for Our Health? Norman Daniels of Tufts University and Bruce Kennedy and Ichiro Kawachi of Harvard argue that income inequality appears to affect health by undermining civil society, eroding social cohesion and political participation.

Photograph courtesy of Stephen J. Suomi, National Institute of Child Health and Development
Richard Wilkinson, a socioeconomic epidemiologist at the University of Sussex (and author of another new book, Mind the Gap), observes that, in wealthy societies, anxiety is one of the most important pathways linking health to social and economic circumstances, because relatively equal societies are less stressful, less hostile and less violent than more unequal societies. Moreover, there is a strong tendency for social relationships and levels of social trust to weaken as the social structure becomes more hierarchical. Wilkinson uses an analogy to primate research on social hierarchy and well-being to draw a clear distinction between the direct effects of social status, on the one hand, and the indirect effects of material circumstances that are closely associated with social status, on the other:
The physiological effects of low social rank were observed in monkeys even in the complete absence of the plethora of differences in socioeconomic circumstances found among humans. The effects cannot therefore be attributed to jobs, housing, smoking, diet, debt, unemployment or whatever. So rather than being linked to social status only indirectly, through such socioeconomic factors, the effects appear to be due to the direct, or "pure," effects of social status.
In other words, direct psychosocial experiences of hierarchy matter; the material circumstances associated with human hierarchy do not.
Taken as a whole, the evidence seems to suggest that there are many factors at work, ranging from the material to the psychosocial, and that it is difficult to assign ultimate primacy to any one mechanism. For instance, living in an environment of tightly constrained choices has both a material and a psychosocial component, as does surviving from day to day on a low income. Loss of control in relation to the material conditions of daily life may contribute to psychosocial stress; on the other hand, strong social support may mitigate it. Finally, it is clear that resiliencies and vulnerabilities can accumulate over the life course. Because of this complexity, George Kaplan, chair of the Department of Epidemiology at the University of Michigan, has called for a shift in emphasis from the search for ultimate primacy to the "epidemiology of daily life."
5. The socioeconomic/psychosocial environment has its impact on health status through the gradient effect on health. There are three fundamental aspects of the gradient, which exerts its effects over the entire life course.
As I mentioned above, the "gradient effect" means that there are successive increases in health status from lower to higher socioeconomic levels in society. This effect has been found in all wealthy societies, and it is found regardless of whether income, education, occupation or a combination of these measures is used to define socioeconomic status. It also appears that the "slope" of the socioeconomic gradient in health is influenced by the level of income inequality in a society, such that the more unequal a society is in economic terms, the more unequal it is in health terms. Moreover, middle-income groups in relatively unequal wealthy societies have worse health than comparable, or even poorer, groups in more equal wealthy societies.
The first notable characteristic of the socioeconomic gradient in health is that it cannot be explained away by reverse causation or differential mobility. In other words, there appears to be a causal relationship between socioeconomic status and health over the life course. The second is that the gradient effect is evident for virtually all of the major diseases that affect health and well-being in our society. Finally, it has been observed that, as the major diseases have changed over time, the gradient effect has replicated itself on the new diseases as they have emerged.
Data from the turn of the 20th century for countries such as England and Wales show the gradient effect for the major causes of disease and death of the time (infectious diseases). Over the next three to five decades those diseases tended to disappear and were replaced by chronic diseases such as heart disease. At first, these new diseases tended to show unique epidemiological patterns, but over time the socioeconomic gradient asserted itself. In the case of heart disease, this shift had taken place by the 1950s. A similar pattern has been emerging over the last 15 years in the case of AIDS.
These characteristics of the gradient have prompted investigators to probe the biological processes that this evidence suggests must connect social circumstances to human resilience, transcending disease-specific processes. An understanding is beginning to emerge as to how early brain development and the development of key host-defense and sensing pathways, such as the autonomic nervous system and the hypothalamic-pituitary-adrenal axis, are co-determined. These links make it plausible that systematic differences in the qualities of human experience will translate into systematic differences in biological responses that will lead, in turn, to systematic differences in health status over time. Those of us working in this field have coined the term "biological embedding" to describe these processes.
Gradient effects appear at the beginning of life in infant mortality and low birth weight. By kindergarten age, gradients are found for cognitive, social and emotional development; they have been measured as (modifiable) socioeconomic inequalities in readiness for school. During early adulthood gradients emerge for mental-health status, obesity and various limiting longstanding illnesses. In mid- to late adulthood gradients are found for a wide range of chronic diseases. Recently, attention has been paid to the mechanisms by which gradients unfold over the life course. Investigators have postulated latent, pathway and cumulative processes.
6. Gradients appear to "flatten up."
Annette deFerrari, Portering/Chickering (Photo Researchers Inc.)
The principal significance of the gradient for policy is simply that gradients can be made to "flatten up." In other words, those societies that produce the least inequality in health and human development across the socioeconomic spectrum have the highest average levels of health and development. International comparisons have shown this for the development of literacy and numeracy skills across the 30 OECD (Organization for Economic Cooperation and Development) member countries and for health status across the European Community. These investigations have shown that "raising the bottom" of the socioeconomic spectrum does not "lower the top," or come at the expense of those at the upper end of the economic spectrum—a fundamental challenge to folk wisdom surrounding economic models currently in vogue. In other words, population health reminds us, first and foremost, that we are a social species and that our health and development depend upon the quality of the social environments where we grow up, live and work.
7. Socioeconomic boundaries to disease around the world are more resilient than geographic boundaries.
Finally, although it is customary to study socioeconomic factors within a nation or society, much new work suggests that the same dynamics work on a global scale. The stories of worldwide tuberculosis and of AIDS in Haiti suggest that it is appropriate, even if difficult, to study how the socioeconomic gradient affects health throughout human society, a forest whose global dynamics we may miss by looking at the trees.
Paul Farmer, a clinician-anthropologist at the Harvard School of Public Health, has worked on the front lines of the fight against infectious diseases in poor countries. His findings illustrate how poverty and other social inequalities can alter disease distribution and sickness trajectories through innumerable and complicated mechanisms, not just those that operate through psychosocial or social-comparison pathways.

David and Peter Turnley/Corbis
Common knowledge always had it that Haiti was an original "point source" and exporter of AIDS to the United States. It was thought that AIDS had spread to Haiti through extensive African contact and been incubated through exotic rites and rituals that spread exposure to human blood throughout the country. Through careful epidemiological analysis, Farmer revealed in the early 1990s what this cultural interpretation concealed. Haiti gradually inherited the North American sex tourism trade from Cuba when Castro eliminated it there after the revolution. There was a large component of gay sex tourism, and Haitian poverty facilitated widespread local participation in the commercial sex trade. Farmer showed that AIDS was imported from North America this way; Haitian direct contact with Africa being virtually nonexistent. The highest rates of AIDS in the Caribbean basin are in the five countries with the greatest trade with the United States, and the lowest rate is in Cuba.
Despite these facts, Haiti was the big loser in the AIDS wars. It was stigmatized as the country that brought AIDS into the Western Hemisphere; its tourist industry, which had become its second largest, was destroyed; and Haitian emigrants to other countries became the object of AIDS-related discrimination. But most important, once AIDS was endemic in Haiti the forces of social inequality could be counted on to turn it into an epidemic. The story of how that happened is a story of factors Farmer labels "structural violence" and "constrained choices." Military dictatorship, crowding, urbanization, migration, loss of peasant lifestyles, lack of effective treatment of other sexually transmitted diseases, gender inequality and institutional inertia all contributed to the process. For example, the 1991 military coup featured intense repression in neighborhoods in Port-au-Prince where the sex trade had been a driving force of the economy. This drove high-risk urban dwellers from urban sex slums back to the countryside, where AIDS had not yet become prevalent.
Most profound, as Farmer claims, his critical re-examination of the Caribbean AIDS pandemic reveals that the distribution of HIV disease does not follow the outline of nation-states but rather matches the contours of the transnational socioeconomic order. Despite the epidemic in Haiti, cases of AIDS are virtually non-existent in high socioeconomic status Haitians. Instead, HIV tracks along steep gradients of international power. Farmer sees one big global gradient in health, wherein the distribution and character of disease among the poor of the world, beset by infectious diseases and violence, is similar regardless of where they live but different from the rich, who face chronic diseases in mid- and late life, regardless of their nation of residence.
In the end, research on the determinants of health reminds us that, despite all the technologies that distance us from the natural world, we are still a social species. The sensing pathways that develop in early childhood and the qualities of the social environments that surround us throughout the balance of our lives combine to create our lived experience. It is this lived experience, more than anything else, that determines our expectation of health. The difference between societies where health expectations are high and those where they are low comes down to the degree to which societies, in their day-to-day living arrangements, honor or violate the basic social character of human existence. No genomic revolution, no matter how profound, will change that basic fact.
Click "American Scientist" to access home page
American Scientist Comments and Discussion
To discuss our articles or comment on them, please share them and tag American Scientist on social media platforms. Here are links to our profiles on Twitter, Facebook, and LinkedIn.
If we re-share your post, we will moderate comments/discussion following our comments policy.