
This Article From Issue
March-April 2026
Volume 114, Number 2
Page 74

Courtesy of North Carolina State University
A surface that has been wiped down with disinfectant will only remain sterile for an instant, before airborne microbes can start to recolonize it. Antimicrobial coatings or materials can help, but they can crack or degrade over time, sometimes releasing polluting microplastics. Richard Spontak, a professor of chemical and biomolecular engineering and of materials science and engineering at North Carolina State University, led an effort to develop a new class of nanostructured polymers that have antimicrobial activity built into their chemical makeups. Contact with these materials will essentially dissolve and break open the membranes of any microbe, and because this activity is not specific to any species or receptor, the chance of a microbe developing resistance to the process is virtually eliminated. Spontak was the recipient of the Walston Chubb Award for Innovation at Sigma Xi’s 2025 International Forum on Research Excellence (IFoRE) and spoke with editor-in-chief Fenella Saunders about his work. (This interview has been edited for length and clarity.)
What got you interested in this area of materials research?
I was keenly interested in marrying together thermodynamics and polymeric materials to get a better handle on how structure forms, and what the structure does in terms of promoting properties. That led up to the discovery of the antimicrobial materials. I had no background in epidemiology, biochemistry, or molecular biology. But it turned out that the materials themselves revealed their properties. They showed us, in an unrelated series of tests, what they could do. It had never been reported before, and it opened up a wonderful door for me to not only do the fundamental work, but also now there was an opportunity to have these materials do something for society.
How does thermodynamics come into play in these materials?
This type of polymer is what are referred to as block polymers. Think of having two or more polymers that have different properties. A polymer in its simplest form can be envisaged as a chain-like macromolecule. If you start taking these single polymers and gluing them together with covalent linkages, you have contiguous sequences of different chemical blocks along the backbone of a single polymer chain. That is what endows these materials with multifunctionality. The most common is to take a glassy polymer and add to it a rubbery polymer, and that makes a thermoplastic elastomer, which is basically a replacement for natural rubber. The thermodynamics are very important here, because they drive the interactions between those sequences and the formation of the structures. In the antimicrobial material, which is an anionic or negatively charged block polymer, we want certain microdomains to be contiguous.
This material is a pentablock polymer, so there are five contiguous sequences within the material. The first and last blocks are glassy. The intermediate blocks are rubbery, and both of those types of blocks are hydrophobic, which is common for most polymers. But the middle block is partially sulfonated, with an added sulfonic acid group, and so it becomes hydrophilic. The molecule itself is what we would call amphiphilic: It can handle oil, and it can handle water.
We want those hydrophilic regions to be contiguous throughout the material. Why? Because once this material becomes hydrated, we want to give the protons on those sulfonic acid groups the opportunity to travel through the material to the surface. There, they form a very thin, low-pH—that is, highly acidic—layer at the surface between the polymer and whatever microbes might come in contact.
How did you discover this antimicrobial property by accident?
My colleagues and I were looking at putting photosensitive dyes that inactivate bacteria into this anionic block polymer. When our student looked at the controls, he found that all of the bacteria were killed. At first, we thought, okay, it was just an experiment gone wrong, maybe there was some contamination. He went back and repeated it probably 10 times and got the same result. We started looking at the topology of the surface, the flow mechanics, or the polymer swelling, but none of those were it. Then we decided that we should take a closer look at the suspending medium. We were shocked to find that the pH was down at about 0.8. At that point we started thinking, is it possible that this material is acidifying the contact layer? We started playing around with the degree of sulfonation and found that, as long as we were above a critical degree of sulfonation, we got the same results.
People have used sulfonated polymers before, especially as antivirals, but they’re always in solution, and they are meant to target certain microbes. Our materials behave in a very different way. They are form-stable, so they don’t dissolve. These materials inactivate microbes very quickly. Five to 10 minutes would be the norm. We can get to the minimum detection limit, typically 99.9999 percent. But I think the aspect of these materials that excites me the most is that they don’t target. Because they are nonspecific in the way that they interact with microbes, it is very unlikely that the microbes could ever develop antimicrobial resistance.
What is the connection between sulfonation and the lowering of the pH on the surface?
Sulfuric acid is a very strong acid, and all the strong acids are infinitely miscible in water. It’s usually introduced in polymers to incorporate some water-loving species. But the one aspect that was never noticed was what effect the sulfonic acid groups had on the surface contact layer. That’s where our antimicrobial studies came into play. Sulfonic acid groups provide those mobile protons, which acidify the surface.
Why does the decrease in pH cause the antimicrobial property?
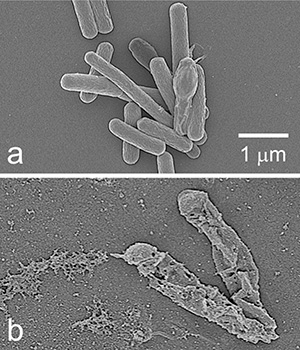
It basically dissolves the microbes. We were able to show that for MRSA (methicillin-resistant Staphylococcus aureus), ampicillin-resistant Escherichia coli, and Clostridioides difficile. That’s why I said it’s non-specific. It basically dissolves pretty much anything that comes in contact, and that includes cells. But it’s much thinner than your outer epidermal layer. So you can touch it for quite a long time. And my suspicion is that the microbes on your fingers would be killed on contact.
How stable is this material?
Polymers, when put into the field, naturally undergo abrasion. If you take a painted wall and you continue to rub it, you’ll see the paint wear thin. When this anionic polymer was put into the field, the biggest issue was abrasion, but we have since solved that problem. We have developed a way to retain the antimicrobial properties and enhance the abrasion resistance considerably, at least over a reasonable lifetime.
Have you completed field experiments?
We do not manufacture these polymers. We turned it over to the original manufacturer, Kraton Corporation. During the pandemic, the Environmental Protection Agency (EPA) granted emergency approval to Kraton to provide Delta Airlines with this polymer as an antimicrobial surface on their ticketing counters and kiosks at several main domestic terminals. If people touched the surface to get their tickets, their fingers were being cleaned at the same time. These lasted quite a while, at least a couple of months.
What is your next research step with these materials?
We have new materials synthesized that we’re sulfonating to different levels to get a better idea of how we can control proton transport. We’re also examining other polymers that aren’t quite as expensive. We are thinking about mobile hospitals, say, in Africa, where they deal with some of the hemorrhagic fever viruses. We’re also now extending out into nonhuman microbes. A lot of the food that we take for granted can be really hurt by microbial infections. We’re turning to eggs and livestock to see if we can prevent these types of infections from occurring.
What is the biggest barrier to getting these materials into widespread use?
The biggest issue right now is just getting permanent approval from the EPA. That was started in 2022, and it’s still not in hand. Part of the problem is that the EPA had no way of classifying these materials. They ultimately classified them as pesticides. That doesn’t really fit at all. But it’s just a hurdle that needs to be overcome. I’m really hopeful that this can be easily used in a number of high-throughput locations, such as hospitals or homes for the elderly. The material could be used as a coating. You could spray this on any surface and it would work.
Could it be used like a paint?
It could also be something simpler. For example, the sprays that we use for disinfectants are a point-in-time solution, but they don’t have to be. A normal cleaning doesn’t last, right? It cleans, and that’s it. Then you have microbial buildup again. As long as you have a solvent that’s not dangerous for people, you could use the same idea and just apply an antimicrobial coating onto a surface that is self-cleaning. It’ll continue to work, and it could last for months.

American Scientist Comments and Discussion
To discuss our articles or comment on them, please share them and tag American Scientist on social media platforms. Here are links to our profiles on Twitter, Facebook, and LinkedIn.
If we re-share your post, we will moderate comments/discussion following our comments policy.