The Penguin's Palette: More Than Black and White
By Daniel T. Ksepka
This stereotypically tuxedo-clad bird shows that evolution certainly can accessorize.
This stereotypically tuxedo-clad bird shows that evolution certainly can accessorize.

DOI: 10.1511/2016.118.36
Color is one of the most important adaptive traits, and one of the best ways to understand it is—improbably—to examine penguins, the quintessential black-and-white animal. The predominant patterns of penguins are accentuated by a subtle but enchanting assortment of colors running the gamut from bold golden yellows to sleek slate blues.

A raft of recent research has revealed that penguins use a surprising array of mechanisms to create color in their beaks and feathers, and has even started piecing together the color patterns of fossil penguin species. Scientists are looking closely at penguins not only to learn more about these fascinating flightless divers, but also to better understand the patterns and boundaries of coloration evolution across all birds.
Penguins provide lessons in color theory, reflecting the artistry of millions of years of evolution. Colors serve many roles in the natural world: camouflaging animals from predators or prey, attracting mates by advertising fitness, and acting as social cues such as conveying juvenile versus breeding status. These roles are not mutually exclusive: Many animals have evolved complex combinations of color patterns that serve multiple functions, and many change their coloration as they mature or through the course of the year. Penguins are no exception. In fact, they exhibit color-generation mechanisms such as novel pigments and unique microstructures that are not seen in any other group of animals. They’ve reinvented blue and yellow hues from scratch, and the rainbow of penguin coloration only increases when we use fossil data to peek into the deep past.
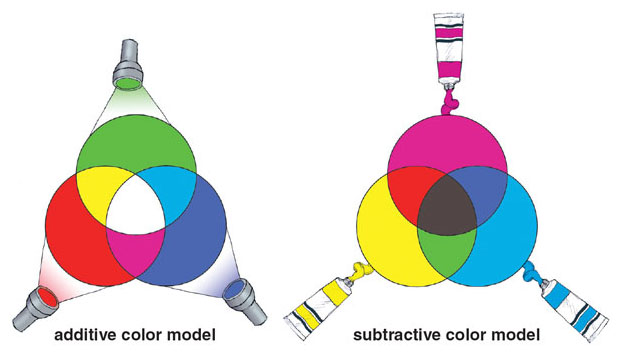
Illustration by Barbara Aulicino.
Color is the property we perceive based on the way an object emits or reflects visible light. This visible light is just one segment of the electromagnetic spectrum, which ranges from the long wavelengths below the radio band to the ultrashort wavelengths of gamma radiation. Humans can see only in the visible light range, corresponding to the red through violet part of the spectrum (approximately 700 to 400 nanometers), but birds and many other animals can detect a wider range.
Interactions between light and biological structures create the colors that we perceive in the natural world. Light itself can have color based on its wavelength, and combinations of light produce additive color. For example, when all frequencies of the visual light spectrum are combined simultaneously, the product is white light. Combining light of different wavelengths can result in different colors; for example, green and red light will combine to produce yellow. Bioluminescent animals such as lightning bugs and jellyfish can produce color in this way.
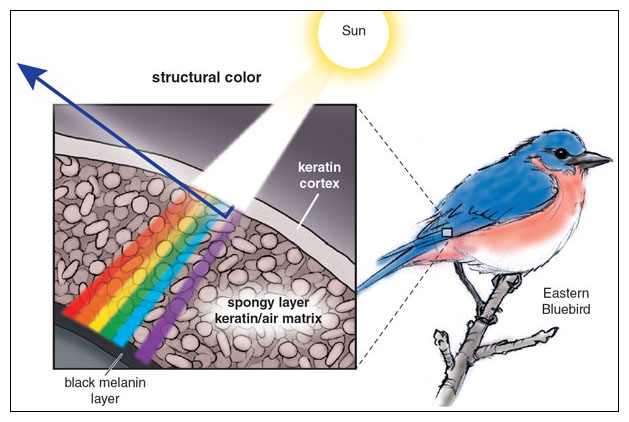
Illustration by Barbara Aulicino.
However, subtractive color is much more common in the biological world. This mode of color generation involves absorption of some wavelengths of light and reflection of others. Pigments create color by absorbing certain wavelengths of light, leaving only the remaining parts of the spectrum visible to our eyes. For example, plants look green because they have pigments that absorb red and blue light.
Finally, structural color is produced by structures that are small enough to interfere with the passage of visible light. Take the example of a bluebird feather, in which microscopic structures reflect blue light while canceling out red and yellow light. Viewed in the sunshine, the feather appears bright blue. However, lighting the feather from behind will remove the reflective effect and cause the same feather to appear dull brown.
Penguins are extreme case studies in the power of shading to perform many of the biological color functions just described. Countershading, when the upper side of an object is darker than the underside, is one of the most common coloration patterns in the animal world. The black-and-white plumage pattern of modern penguins is a classic example of countershading: The dark upperparts help obscure the penguin when viewed from above against the dark seafloor, and the light underparts likewise obscure it when viewed from below against the light filtering in through the water.

Doug Allan (above), Bryan and Cherry Alexander (below)/Nature Picture Library
Of course, this black-and-white pattern makes penguins highly visible on land, especially when they are milling about on ice. However, sacrificing obscurity on land for camouflage in the water makes sense for penguins. They do all of their hunting underwater, and the predatory sharks, leopard seals, and orcas that they fear most are aquatic as well. Adult penguins tend to see the land as a safe place, which is in large part due to the fact that they molt and nest in areas with few or no terrestrial predators. Thus, conservative black-and-white plumage predominates across most of the penguin world.
Tuxedos are fine, but why not spice things up with some brightly colored accessories? One reason to add a dash of color is sexual signaling. Many animals advertise their fitness or breeding status to others of their species. Producing bright colors can be costly in two ways, both metabolically (in terms of nutrient cost to generate the colored structures) and ecologically (making an individual more obvious to predators). Flashing a bright patch of feathers sends the message that a bird is in such good condition that it can afford the cost. Penguins cannot don cummerbunds and novelty bowties, but they have evolved many striking accessory color patterns that span the rainbow in hue.
Black-and-white penguin tuxedos fulfill the animal’s need for camouflage; small splashes of color handle sexual and species signaling. But the actual execution takes place through a wonderful, startling variety of mechanisms that reveal the intricate process of evolution. Crested penguins are graced with golden head plumes. This group includes such favorites as macaroni penguins, royal penguins, and rockhopper penguins. Each species has a distinct arrangement of golden head feathers: Length, hue, and orientation help distinguish the different species. The yellow-eyed penguin is a close relative of the crested penguin group. One of the most amazing sights in the avian world is this bird’s eponymous bright yellow iris. Yellow-eyed penguins lack head plumes, but are equipped with a band of yellow feathers that surrounds their eyes like a miniature mask of Zorro.

Andy Trowbridge/Nature Picture Library
Where does this burst of color come from? Many birds have yellow feathers, but they create this color in different ways. It may come as a surprise to find that some birds with yellow feathers cannot synthesize the color on their own. Canaries, epitomes of yellow birds though they are, must obtain the pigment from their food. They do so by extracting carotenoids, orange or yellow plant pigments, from seeds. These pigments provide bright colors to many familiar backyard birds, such as the brilliant red of cardinals and the dazzling yellow of goldfinches.
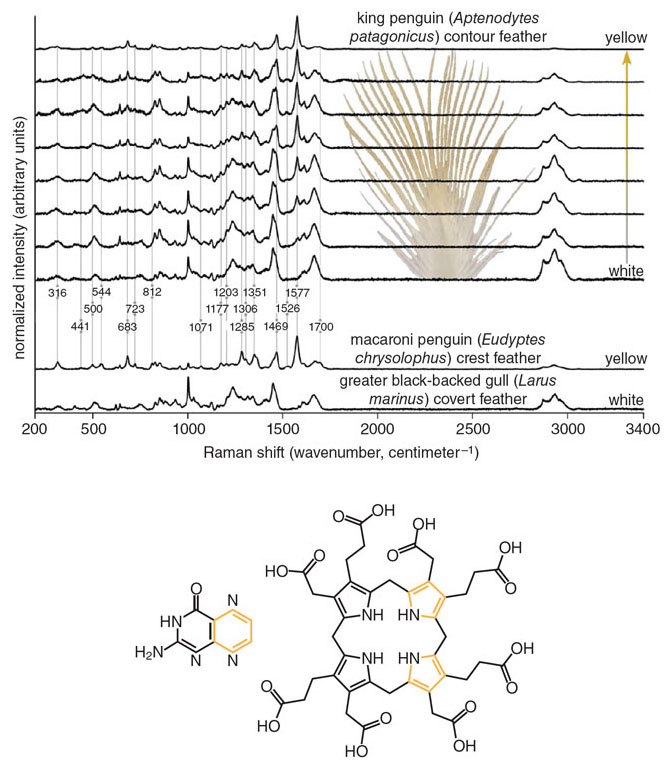
Image from D. B. Thomas, C. M. McGoverin, K. J. McGraw, H. F. James, and O. Madden, Journal of the Royal Society Interface, 10:20121065.
Penguins do not need any help from plants to create yellow feathers. This is just as well, because their appetites tend toward fish and krill, rather than seeds and berries. It turns out that the pigment responsible for yellow color in crested penguins is completely unique among birds. In 2013, a team of ornithologists and chemists led by Daniel Thomas, currently at Massey University in New Zealand, used Raman spectroscopy to bounce lasers off penguin feathers and were surprised to find a chemical signature unlike that of any known pigment. The Raman spectrum observed in the penguin feathers shares some characteristics with porphyrins, a type of pigment formed from amino acids and found in birds such as the brightly colored turacos. The team named the new pigment spheniscin after the scientific name for the penguin family, Spheniscidae. However, further work is needed to clarify the molecular structure and formal chemical name of the pigment. One thing is certain: Penguins are able to create spheniscin themselves, as demonstrated by captive penguins maintaining their yellow hues regardless of their diet.
Golden hues make for dashing penguins, but these fancy colors nevertheless come at a cost—in energy and risk of predation. To crested penguins, this rakish set of yellow head plumes is worth that cost. Top-condition plumage signals healthiness to potential mates. Ecological experiments have shown that giving a penguin a “haircut” that removes the yellow feathers causes its chance of mating to plummet. The plumes seem to declare, “I am such an excellent penguin that I can afford to build and flash these fancy plumes” (the penguin equivalent of a boastful Tinder profile picture).
The new and exciting ability to reconstruct color from fossils recently revealed that penguins’ history may be more technicolored than was once assumed. Original biological color is almost always lost in the fossilization process. Thus, a feather that may have been bright red in life may end up as a fossil with a charred blackish color due to degradation. For a long time, this left paleontologists unable to do more than guess at the colors of extinct species. In 2008, a team of researchers led by Jakob Vinther, currently at Bristol University, honed in on a microscopic signal of fossil color: the miniscule but mighty melanosome.
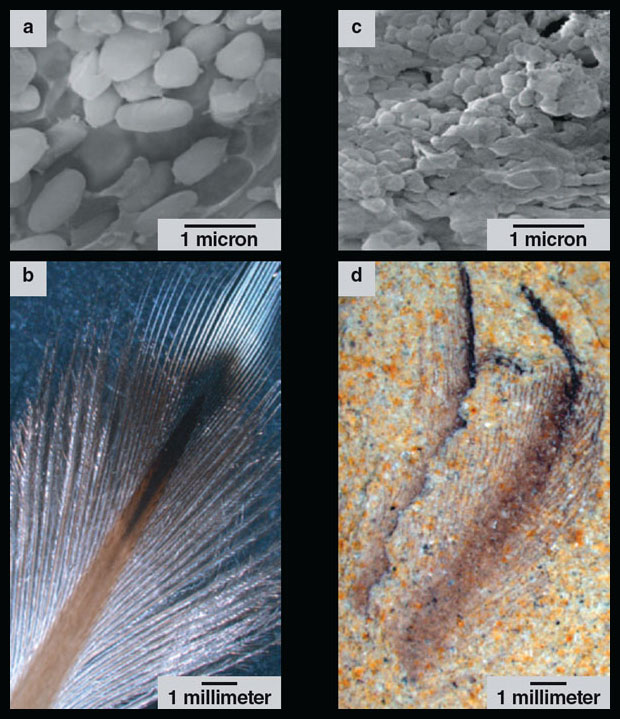
Images from J. A. Clarke, et al., Science 330:954.
Melanosomes are organelles that bear melanin, a pigment that plays a role not only in coloration but also in protecting cells from harmful ultraviolet radiation. Melanosomes occur within many biological structures such as feathers, human hair, vertebrate eyes, and even squid ink. Averaging just a single micron in length, melanosomes are invisible to the naked eye. In fact, their small size caused them to be overlooked in the fossil record for many years: Clusters of melanosomes in fossil feathers were long mistaken for bacteria.
Melanosomes are tough. In general, “hard parts” with a mineralized component such as bone or shell have good odds of making it into the fossil record, whereas “soft parts,” such as muscle, blood vessels, and skin, degrade rapidly after death. Melanosomes are an exception: They are resistant to bacterial decay, chemical decomposition, and physical deterioration. In fact, if you are a birdwatcher you may have already witnessed the tenacity of melanosomes firsthand by observing the changing plumage of birds such as gulls over the course of the year. Black-and-white banded feathers tend to wear unevenly because the melanosome-rich dark regions resist deterioration better than the white regions where melanosomes are absent. Thus, a late-season gull with differentially worn feathers will have a darker appearance then a freshly molted gull.
Although intact melanosomes do not result in fossil feathers retaining life colors, they do provide a signature to reconstruct original coloration, because different types of melanosomes bear different pigments. Eumelanosomes, which contain a pigment called eumelanin that produces black and gray colors, look like hot dogs under a microscope. Pheomelanosomes, which contain the pigment pheomelanin that produces reddish browns and yellows, look more like miniaturized M&M candies. Thus, the aspect ratio (proportion of length to width) is the key to telling what color pigment a melanosome originally carried. Scientists can measure fossil melanosomes using scanning electron microscopes and compare their proportions to those of modern melanosomes to reconstruct the colors of extinct organisms.
In 2010, this method was tested by a team led by Julia Clarke of the University of Texas at Austin after a magnificent 36-million-year-old penguin fossil was unearthed in Peru. This fossil comes from the middle stage of penguin evolution, roughly halfway between the age of the oldest known penguin fossils (62 million years in age) and the modern penguin radiation. Nicknamed “Scaly Pedro,” because its foot scales were intact, the specimen also preserved the first ever fossil penguin feathers.
The excitement was palpable, as I can attest, having been a member of the research team that first exposed the fossil from the rock matrix. Our team decided to give Scaly Pedro the formal name Inkayacu paracasensis, which means “water king.” When the fossil feather samples were analyzed at a scanning electron microscope facility, intact melanosomes were identified within fossil feathers from the flipper and from isolated contour feathers. One could be forgiven for expecting they might be black eumelanosomes. Instead, we were surprised when feathers from the underside of the flipper preserved phaemelanosomes indicating a reddish-brown coloration, and body feathers yielded evidence for both gray and reddish-brown colors. These colors are seen in some juvenile penguins today, but the skeleton of I. paracasensis indicates it was a full-grown adult.

Illustration by D. W. Miller.
What was I. paracasensis doing with its unusual colors? One possibility is that this species inhabited different environments than typical modern penguins and thus moved away from a standard black-and-white countershading strategy. While countershading prevails in many marine predators such as orcas and sharks, other animals such as seals show more even coloration. Perhaps I. paracasensis foraged in murkier coastal waters, rich in runoff from adjacent rivers, and thus employed a more evenly colored coat of feathers. This idea raises the question of whether such coloration was a primitive pattern for penguins that was later replaced by countershading, or if instead I. paracasensis represents a side branch of the penguin tree experimenting with a new coloration strategy.
Ancient penguins were not black and white, and some modern penguins are not, either. As its doubly informative moniker implies, the little blue penguin is both the smallest species of penguin (barely tipping the scales at just 2 pounds) and is also cloaked in a coat of slate-blue feathers. Little blue penguins form colonies and nest in burrows and under vegetation, sometimes even digging down under people’s sheds. One of the most wonderful sites in the natural world is a “raft” of a few dozen little blue penguins coming ashore en masse, an evening ritual at their colonies as the tiny adventurers return from a day of foraging at sea.

Wikimedia Commons
Not all penguin colors are produced by pigments, and little blues provide an exciting example of structural color. In birds, there are many dazzling examples of structural colors produced by the way feather components scatter and split light. Blue is one of the most striking structural colors, and has attracted much interest from biologists. β-keratin, a protein found in scales, claws, beaks, and feathers, is often involved in generating blue. In the feathers of some species, such as bluebirds, the color is created by miniscule spherical or channel-shaped air bubbles forming within the β-keratin matrix of the feathers. Blue also occurs in the skin of many birds, such as the helmeted guineafowl. The blue facial skin of that species is another example of structural color, in this case created by a thick layer of parallel collagen fibers that results in coherent scattering of light.
In 2009, Liliana D’Alba of the University of Akron and her colleagues took a deep look at little blue penguin feathers and were surprised to find that they utilize a previously unknown mechanism to create blue coloration. Unlike other blue-colored birds, these pint-sized penguins produce miniscule β-keratin nanofibers arranged in tiny parallel bundles to generate blueness. These bundles are packed inside the medullary cells of their feather barbs, sandwiched between an outer cortex of unstructured β-keratin and an inner layer of cortical cells containing solid β-keratin and clusters of melanosomes (see figure below). The nanofibers are truly tiny—about 1/5000 of a millimeter in diameter. This small size allows them to interfere with light waves. Blue light is refracted by the nanofibers, whereas other colors of light are absorbed by the layer of melanosomes below.
These novel nanofibers reveal that penguins essentially re-evolved the color blue, independently from other types of birds. Moreover, they may have found an efficient way to do so: Other types of β-keratin fibers are known to self-assemble without requiring energy from cells, and it is likely that the nanofibers involved in blue penguin feathers do so as well.

Image from L. D’Alba, et al., Biology Letters 7:543.
Blue seems to be a popular color for birds—collagen-based blue skin coloration is believed to have evolved more than 50 times independently. Scientists speculate that the repeated evolution of blue skin in birds may be due to the inherent nature of collagen. In their role of supporting skin, collagen fibers are organized parallel to the surface in the facial region and spaced closely enough to be near the distance to interfere with wavelengths of visual light. Mutations resulting in enough collagen reorganization to generate blue color appear to have arisen relatively frequently. “Bluer” birds seem to have been more attractive to potential mates, resulting in the trait becoming fixed in dozens of separate bird lineages. The repeated exaptation of collagen for generating blue makes the unique nature of the β-keratin nanofibers in penguins all the more interesting. Penguins did not take one of the established routes to blue, but carved out their own evolutionary pathway. Perhaps other methods of creating blue await discovery even now.
Some of the most interesting penguin colors are the ones that the human eye cannot see. Tall in stature and long of beak, king penguins bear striking color patches along their necks, ear regions, and the sides of their beaks. In the visible light spectrum, these beak patches appear orange. Penguins are able to see into the ultraviolet region of the electromagnetic spectrum, however, and can detect additional patches of ultraviolet color along the lower bill that are imperceptible to human eyes.

Michael Nolan/© robertharding/Alamy Stock Photo Bottom image from B. Dresp, P. Jouventin, and K. Langley, Biology Letters 1:310.
How do these penguins produce ultraviolet colors? In this case, the answer is another type of structural color. King penguins, as well as their more famous relatives the emperor penguins, have evolved a multilayered reflector photonic microstructure that reflects ultraviolet light. A multilayer reflector is composed of alternating layers of high refractive index and low refractive index materials. Reflected light from the different layers can interact to produce different colors, and this mechanism has been observed in colors ranging from metallic golds in beetles, to silvery scales in fish, and now also to the ultraviolet markings of penguins.
Close study by Birgitta Dresp-Langley of the University of Strasbourg and her colleagues revealed that the outer layer of tissue surrounding the beak in king penguins contains special folded microstructures with intervening filaments of β-keratin. Under a transmission electron microscope, these structures could almost be mistaken for ripples on the bottom of a sandy stream. Once again, penguins seem to have evolved their own mechanism for creating color. The microscopic structure of the tissue creating the ultraviolet markings in king penguins appears to be arranged by a merging of membranes. Adjacent cell membranes are hypothesized to interdigitate in order to form the elaborate folding microstructures that result in reflectance.
As with the plumes of crested penguins, scientists hypothesize that these beak markings signal sexual maturity and make penguins more attractive to mates. As a king penguin matures, the ultraviolet hue of the beak markings increases. In wild penguin studies, scientists have documented higher ultraviolet reflectance in recently formed male–female pairs. To put the attractiveness hypothesis to the test, researchers obscured the ultraviolet markings of some male penguins by covering them with a coat of varnish. They then observed that penguins with covered markings took longer to find mates than their peers. This observation suggests that penguins with less bold ultraviolet marks were less attractive to females.
Penguins have already revealed a treasure trove of new data for understanding avian color evolution. Scientists will continue to push deeper into the world of penguin beaks and feathers in coming years, and these analyses have the grand potential for reciprocal illumination. Tracking down the physical and chemical details of color-producing tissues in penguins expands our understanding of the range of ways that birds can produce color under different evolutionary constraints, such as diet, environment, and ecology. In turn, mapping these coloration strategies onto the evolutionary tree of penguins reveals the deep and secret history of how a return to the oceans influenced the evolution of these remarkable birds’ camouflage, feather strength, and breeding plumage. As we keep pushing into the microscopic world of penguin color, who knows what we will find next?
Click "American Scientist" to access home page
American Scientist Comments and Discussion
To discuss our articles or comment on them, please share them and tag American Scientist on social media platforms. Here are links to our profiles on Twitter, Facebook, and LinkedIn.
If we re-share your post, we will moderate comments/discussion following our comments policy.