Taste, Sickness, and Learning
By Terry L. Davidson, Anthony L. Riley
Understanding how we form aversions to particular flavors has led to new ideas about learning—and could have implications for treating obesity and drug use.
Understanding how we form aversions to particular flavors has led to new ideas about learning—and could have implications for treating obesity and drug use.

DOI: 10.1511/2015.114.204
Imagine that you are dining at a familiar restaurant, and you order a new item on the menu—something that you’ve never tried before—and later that night you become violently ill. What caused you to get sick?
Your illness could have been caused by a touch of the flu, a familiar food that was poorly preserved or prepared, an exposure to a toxin, or a favorite cocktail interacting badly with some medication taken earlier in the day. But even if you are aware of these and other alternative possibilities, there is a high probability that you will blame the novel dish for your illness. Indeed, the taste, and even the thought, of that new menu item may subsequently make your stomach turn, and you may decide never to eat that food again.

OJO Images Ltd/Alamy
No doubt many of us have had this type of experience. Why are we so quick to place the blame for sickness on a novel-tasting food instead of blaming many other equally plausible possibilities? You may be thinking that blaming the unfamiliar food is the most logical response, but why does it seem that way? We’ve eaten new things many times before without becoming ill, and we’ve become ill before without eating anything new. What makes the connection between a novel taste and illness so strong that it can override these other types of experiences?
Answers to these questions, as well as evidence for the reality of the phenomenon itself, were found not in anecdotes but in the results of experiments. Those results shook the foundations of psychology as it existed at the time, and led to a paradigm shift in thinking about how humans and other animals learn in general, and about the conditions under which learning occurs. Our group and others are still exploring the implications of those findings today.
Studies of what would later be termed conditioned taste aversions date back to at least the 1940s. One of the first of these investigations was conducted in support of the British war effort by Charles Elton of the Bureau of Animal Population. In an attempt to develop procedures for the eradication of rats and mice from foxholes and beachheads, Elton and his fellow researchers observed the consequences when rats and mice consumed poisoned baits. In these studies, the rodents would often sample the baits but they would eat only small amounts. This reluctance served to protect the rodents from death, but did not spare them from a rather intense poison-induced illness. After recovering from this toxicosis, the rodents stopped consuming the poisoned baits. This same strategy in the field would have the rats and mice avoiding foods associated with poison, but they would persist by living off other available foods. Julian Rzóska, one of the investigators in the research effort, surmised that the animals learned that the base food in which the poison was embedded made them sick and then they avoided the food on subsequent exposures.
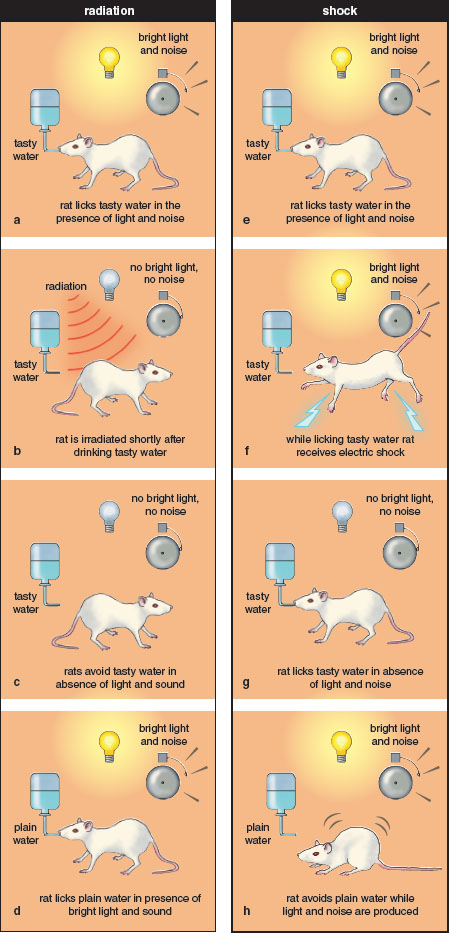
Illustration by barbara Aulicino.
In the 1950s, John Garcia demonstrated conditioned taste aversion under quite different laboratory conditions and came to realize that the phenomenon represented much more than a potential means of improving pest control. Early in 1951, Garcia left graduate school at the University of California, Berkeley, and began working at the US Naval Radiological Defense Laboratory at Hunters Point in San Francisco, where he used the rat as a model to study the effects of exposure to radiation on living systems. He noted that rats given water in plastic bottles prior to the induction of radiation sickness subsequently avoided drinking water from those bottles. The same rats would drink the water if it was provided in glass bottles. Garcia and his colleagues speculated that the plastic containers may have given the water a novel taste that the rats associated with the subsequent sickness. Garcia tested this idea by giving rats a novel sweet-tasting saccharin solution during radiation exposure. As expected, when the irradiated rats were tested later, they strongly suppressed intake of the saccharin solution compared to a control group that had not been irradiated after consuming saccharin. The aversion to saccharin persisted for over a month despite the fact that it was acquired after a single pairing of saccharin with radiation.
An observation that immediately struck Garcia was that the rats seldom avoided the compartments in which radiation was delivered, and if they did, such avoidance took longer to establish and was less stable than taste aversion. The important implication of this observation was that not all stimuli (in this case, external environmental cues versus taste cues) were equally associable with radiation sickness.
In a landmark test of this hypothesis, Garcia and his colleague Robert Koelling conducted a study in which licking a spout not only delivered a saccharin solution but also activated a flashing light and a clicker. During exposure to this “tasty/bright-noisy water,” the rats were irradiated. In the test phase, animals that were given the tasty/bright-noisy water compound paired with irradiation avoided consuming the saccharin solution when it was subsequently presented without the flashing light and clicker. However, the same rats continued to lick the tube when it no longer contained saccharin, but licking still produced the light flash and click. These results showed that sweet taste was much more strongly associated with radiation sickness than were the light and clicker even though pairing with illness was equated for all three types of stimuli.
Interestingly, when these same stimuli were paired with electric shock in other groups of rats, only the audiovisual cues, and not the sweet taste, acquired the ability to suppress licking. From these results, Garcia argued for a selectivity of learning in which tastes were preferentially associated with sickness and auditory and visual cues were preferentially associated with shock. That is, learning not only depended on pairing a cue with a consequence, it also depended on which specific types of cues were paired with which specific types of consequences. This conclusion contrasted with the prevailing views, which considered all types of stimuli and consequences to be equally associable.
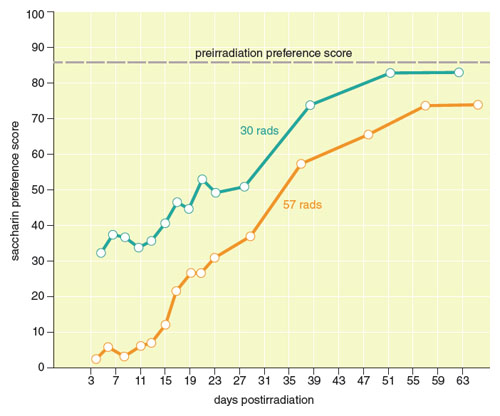
Data adapted by Barbara Aulicino from J. Garcia et al., 1955..
Garcia and his colleagues confirmed another atypical characteristic of taste aversion learning when they showed that rats given saccharin to drink followed by drug-induced illness (vomiting caused by an injection of apomorphine) acquired an aversion to the saccharin solution even when the injection was delayed by as long as 75 minutes after consumption of saccharin. Such long-delay learning also contrasted sharply with the widely held view, based on studies in which auditory or visual cues were paired with food or shock, that little or no learning would occur with delays greater than seconds.
The findings that strong taste aversions could be acquired after only one pairing of a taste with illness, that tastes compared to other external cues seemed to be selectively associated with illness, and that taste-illness associations could be formed over long delays, violated what others believed to be the laws of learning. Garcia further antagonized the largely behaviorist establishment at the time by arguing that evolution had shaped these characteristics. Specifically, the ability to associate a taste with the effects of poisoning after a single exposure was highly adaptive in that it enabled animals to avoid the potentially fatal consequences of repeated sampling of toxins. Given that illness is often produced by toxic plants or decaying meat, animals that were able to selectively associate tastes with illness were at an evolutionary advantage. And chances of survival were also increased by the ability to bridge temporal gaps between ingestion of foods and the delayed onset of illness that resulted from the slow absorption of toxins during the normal digestive process.
Prevailing learning theories made no accommodations for such adaptive specializations. As a result, taste aversion learning and its interpretation were met with skepticism. For example, Garcia’s finding of selective association (the bright/noisy water experiment) was rejected by several journals, as were several of his other initial findings. Garcia later handled such reactions quite humorously, calling editors and reviewers “neophobic creatures” that were reacting more to the novelty of his findings than to their scientific merit or theoretical implications.
Although such reactions slowed the acceptance of Garcia’s work, the robustness of conditioned taste aversion learning and its replicability by other researchers helped to overcome this initial reticence. Moreover, the challenges to learning theory initially posed by Garcia’s findings and ideas soon extended well beyond conditioned taste aversion. Data from many laboratories began to accumulate that showed that behaviors could be learned either very quickly or only with great difficulty, if at all, dependent on how well the task parameters and response requirements matched a given animal’s natural history and behavioral repertoire.
With so many examples from other domains, taste aversion learning could no longer be viewed as an anomaly. Instead, it became the foundation for the fledgling view that there are biological constraints on learning. This view proposed that although learning itself is a general phenomenon, it could be constrained or facilitated based on the natural history of the animal, and that theories of animal learning and conditioning had to take that natural history into account. Thus, taste aversion learning lost its unique status and was seen instead as one of many different learning phenomena that demonstrated a fundamental role for evolution in behavior.
Although it was clear that evolution forged fundamental learning processes that were shared by many species, it was also recognized that the details of learning for any individual species could differ based on the demands encountered during that species’ evolutionary past. The blending of biology and psychology has led to the widespread acceptance of the idea that behavior is the product of an interaction involving both an animal’s individual (ontogenetic) and species (phylogenetic) histories.
Beginning in the middle of the 1970s, principles that had emerged from the earlier studies of taste aversion learning were applied to a variety of other issues. That is, taste aversions were not being studied only to further characterize the phenomenon but also to explore ways in which it could be used to understand and treat a host of other behavioral issues. Such applications have included its use in studying acquired food aversions and preferences during pregnancy, changes in food preferences accompanying cancer development, the biological and neurochemical mediation of learning and memory, the control of attacks on domesticated animals by natural predators, the nature of and control of immunosuppression, and the side effects associated with chemotherapy.
The work of Ilene Bernstein and her collaborators at the University of Washington in the late 1970s provides an important example of this latter application. Bernstein noted that children undergoing chemotherapy often acquired aversions to the food they consumed around the time of drug treatment. To substantiate these informal observations, Bernstein gave children a novel food (a unique-tasting ice cream) immediately prior to receiving chemotherapy and found that they decreased their consumption of the ice cream when they had the opportunity to consume it again, compared to children that received no chemotherapy or that received chemotherapy that was not paired with the ice cream.
Chemotherapy had less impact on children's normal diet when a novel food was given during a session.
This result showed that the pairing of the novel ice cream with chemotherapy was sufficient to condition an aversion and provided an explanation for why children undergoing chemotherapy exhibited changes in food preferences. Importantly, work from other animal models gave insight into ways to reduce or eliminate such aversions and ameliorate this often-reported side effect of chemotherapy. Specifically, novel foods had been reported to have a higher likelihood of being associated with illness than more familiar ones. This very same premise was used by Bernstein and her colleagues to demonstrate that chemotherapy had a lessened impact on the children’s normal diet when a novel food was given during the chemotherapy session. In other words, when the children ate a novel food such as the ice cream prior to therapy instead of a familiar one, there was an increased likelihood that the novel food, and not foods from their normal diet, would be associated with the illness. This creation of a dietary scapegoat precluded the avoidance of essential and familiar components of the child’s diet.
The same mechanism of forming a food aversion from pairing a specific taste with gastrointestinal illness turns out to have a parallel positive outcome of forming food preferences, when a taste or flavor is paired with a nutritive or caloric boost in the digestive system. This latter type of learning has been convincingly demonstrated by Anthony Sclafani and his associates at Brooklyn College-CUNY. That team used what Sclafani termed the “electronic esophagus,” which paired the oral intake of a non-nutritive flavor with gastric infusion of a nutrient (a fat or carbohydrate solution) that completely bypassed the mouth, yet yielded a strong learned preference for the paired flavor. This outcome does not seem very different from the formation of a food aversion produced by pairing oral consumption of a non-nutritive taste with the intraperitoneal injection of a toxin, in that both the preference and the aversion are based on pairing orally consumed stimuli with post-ingestion consequences. Moreover, just as learned food aversions can be seen as performing an adaptive function by protecting animals from ingesting harmful substances, food preference learning can be seen as adaptive by promoting intake of substances that are needed to sustain life. So, we wondered, what would be the consequences of interfering with each of these adaptive functions?

Images courtesy of Bill Given/The Wild Source.
If one reduced the ability of animals to use the taste of a toxic substance to predict subsequent illness, this result would presumably have adverse consequences for survival by increasing the likelihood of poisoning. Likewise, research we did in collaboration with Susan Swithers and her research group at Purdue University suggests that reducing the ability of tastes to predict caloric outcomes may also produce harmful, but perhaps less immediate, effects on survival. For example, it has often been proposed that based on the normally strong association between sweet taste in the mouth and the arrival of calories in the gut, sweet tastes acquire the capacity to evoke a number of preparatory physiological responses that promote efficient energy use. Given this function, one might expect that experiences that weaken the sweet taste–calorie association should also interfere with energy and body weight regulation. One way to weaken the adaptive sweet taste–calorie association would be to consume noncaloric sweeteners, which would involve exposure to sweet taste that is not followed by calories. A variety of experiments with rats have provided support for this hypothesis by showing that exposure to noncaloric sweeteners is followed by increased weight gain and weaker control of the intake of caloric sugars. A 2014 review by Swithers shows that similar adverse effects of consuming noncaloric sweeteners have been reported for humans. Thus, both rodent and human data suggest that obesity and its associated problems may be adverse health consequences of weakening the sweet taste–calorie association.
The tongue is not the only part of the body with the ability to taste, and hence to play a role in taste aversion. Recent findings establish that basic taste stimuli (such as sweet and bitter) are detected in the gastrointestinal tract using taste receptors and chemosensory signaling pathways that are much the same as those that are used by the tongue. Andrea Tracy, and later Lindsey Schier, led investigations at Purdue University that provided evidence that gastrointestinal tastes, like orally detected tastes, can be associated with toxicosis, and that those associations could alter ingestive behavior.
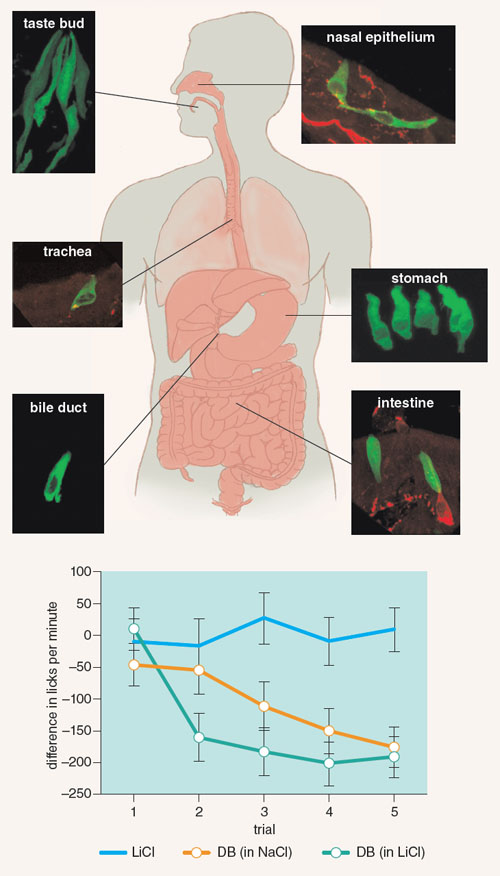
Illustration adapted by Barbara Aulicino from R. E. Finger and S. C. Kinnamon, F1000 Biology Reports 3:20. Micrographs courtesy of Marco Tizzano. Graph data adapted from L. A. Schier et al., 2011.
Tracy first confirmed Sclafani’s earlier findings that rats learned to prefer an orally consumed non-nutritive flavor (such as orange and cherry) that was paired with infusions of nutrients (carbohydrate or fat emulsions) directly into the gastrointestinal tract, relative to flavor cues that were paired with water, despite the fact that the delivery of the nutrient emulsion completely bypassed the oral cavity. She then paired one combination of oral flavor and intestinal nutrient infusion (such as cherry and fat) with illness induced by injections of lithium chloride (LiCl), and the other compound (such as orange-carbohydrate) with saline injections as a control. As expected, intake of the oral flavor paired with LiCl was reduced compared to the flavor paired with saline when those flavors were presented without nutrient infusions. But in addition to this standard conditioned taste aversion, when given the opportunity to consume the gastrointestinal-infused nutrients by mouth without the flavors, Tracy also found what appeared to be an intestinal taste aversion. Specifically, the rat’s preference for the LiCl-paired nutrient emulsion was significantly reduced compared to the saline-paired emulsion, despite the fact that neither nutrient had made contact with oral taste receptors during pairing with LiCl. Apparently, what was learned about nutrients detected solely in the gut modified the ingestive behavior of the rats when they had their first opportunity to orally consume each nutrient. She subsequently showed that rats could learn not only about nutritive intestinal stimuli but also about non-nutritive intestinal flavor cues, if those flavors had been previously infused into the gut in compound with nutritive solutions.
Schier extended these findings by establishing intestinal taste aversion learning in studies that assessed rapid, moment-to-moment changes in ingestive behavior. In one experiment, thirsty rats were trained to lick at a sipper spout for a solution of table salt (sodium chloride, NaCl). A brief intestinal infusion of either the same salt, or one of the same concentration made with a toxic LiCl solution, was yoked to their licking behavior for the first 6 minutes of each 30-minute session. Because rats were licking for the same NaCl solution at the sipper spout when either NaCl or LiCl was intestinally infused, oral taste cues could not predict the LiCl infusion. Therefore, the rats were not able to predict subsequent malaise by differentiating the intestinal infusion of LiCl from NaCl during the early 6-minute infusion period. However, when bitter-tasting denatonium benzoate was laced into the intestinal infusion of LiCl, the rats learned after a single trial to rapidly curb intake within minutes of the infusion, thereby reducing further accumulation of the malaise-inducing LiCl.

Wikimedia Commons
Intestinal infusions of non-nutritive sweet tastes delivered with LiCl could also reduce ongoing intake if those sweet tastes had previously been mixed with nutritive sugars. Thus, this study showed that intestinal taste aversion learning about an intestinally infused non-nutritive sweet taste could be prompted by training it in conjunction with a nutritive sweetener.
This series of experiments confirmed that taste stimuli are detected by receptors located in the gastrointestinal tract; provided new evidence that these gastrointestinal tastes can be associated, much like oral taste stimuli, with gastric malaise; and the association of gastrointestinal taste cues with malaise can alter both food preference and ongoing ingestive behavior.
Associations between intestinal tastes and either aversive or nutritive post-ingestion consequences could have several functional roles in feeding behavior, such as providing a last line of defense prior to the absorption of significant amounts of toxins, promoting learning about oral taste stimuli by filling the temporal gap between the detection of oral tastes and postingestive outcomes, or contributing to control of digestive processes that may occur well after oral consumption has ended.
It does not seem likely that this intestinal taste system evolved as a protective mechanism that operates independently of oral taste. Taste stimuli do not usually arrive in the gut without first passing through the mouth. However, there are conditions where this does happen. Tube feeding and the use of medicinal capsules are two ways in which tastes could be detected in the gut while bypassing oral taste receptors.
One implication of the above findings is that if these routes of administering nutrients or medications are associated with gastrointestinal malaise produced by other sources, the tastes of the intubated or encapsulated substances could be associated with the illness and thus avoided upon subsequent oral contact. This outcome is presumably less problematic when capsules are used to contain bitter tasting substances that would not be well tolerated if consumed orally. However, tube feeding provides nutrients and thus the associating intestinal taste of these nutrients with illness might negatively impact their subsequent consumption by mouth after tube feeding is suspended. The implications of this possibility for medical interventions merit additional study. One speculative suggestion is that by adding bitter taste to intubated nutrients, the association of the intestinal tastes of the nutrients with illness might be reduced or prevented. Although this approach could increase aversion to bitter tastes, it would have minimal impact on subsequent oral intake, because bitter tastes are usually avoided when consumed by mouth even in the absence of association with illness.
Although the initial work on taste aversion learning focused on illness as the effect that conditioned such avoidance, in the early 1970s, a variety of researchers studying taste aversion learning reported that drugs known to be rewarding in certain contexts were also quite effective in suppressing the intake of fluids with which they had been associated, likely because of their ability to cause side effects such as anxiety, vomiting, or drowsiness. The ability of drugs to be both rewarding (supporting self- administration) and aversive (inducing a taste aversion) was initially considered paradoxical, or thought to be a function of very different conditions (such as different doses) used to establish each behavioral effect. Further research, however, indicated that drugs such as morphine, cocaine, amphetamine, and tetrahydrocannabinol (found in marijuana) were complex compounds with multiple stimulus effects and, depending upon the behavior being investigated, could induce aversions or preferences.
Data adapted by Barbara Aulicino from J. Cobuzzi et al., 2013.
This ability had interesting implications for how these effects might interact in any specific individual to affect drug intake. If drugs such as morphine have both positive and negative effects, then this balance must play a role in determining the drug’s use. Importantly, factors that determine each of these affective properties would, in turn, influence the relative balance of the two, and thus increase or decrease the drug’s potential for abuse.
One such factor that we have investigated with our collaborators at American University in Washington, DC, is drug history. This work has focused on how history with a drug (whether that be prenatal, during maternal rearing, as adolescents, or as adults) affects the subsequent rewarding and aversive effects of a drug. Most research on this topic has been on how drug exposure impacts the drug’s rewarding effects. For example, if an animal develops tolerance to such effects, it may increase intake to compensate for the decrease in the drug reward.
Data adapted by Barbara Aulicino from J. Rinker et al., 2011.
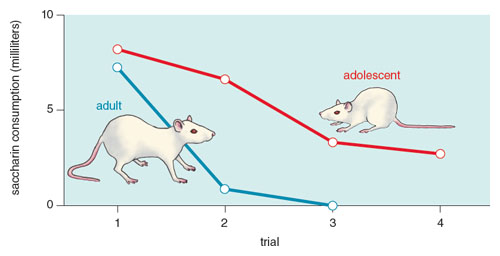
One can also ask how changes in the drug’s aversive effects can alter drug intake. In this case, a rat’s continued drug use during adulthood significantly decreases the drug’s aversive effects as the animal adapts to the specific properties of the drug, similar to an adaptation that one finds in heroin users and tobacco smokers who report nausea when initially taking the drug, but not after using it continually for some time. Indeed, drugs that in naive animals can induce significant and robust aversions have little effect in animals pre-exposed to the drug. This pre-exposure effect can be evident even when the two drugs are different (say, alcohol and cocaine).
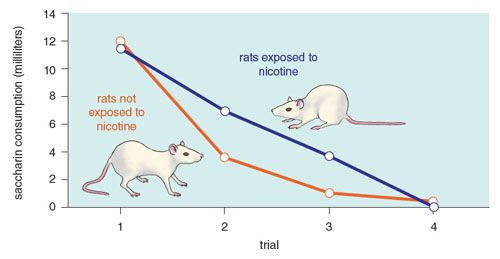
Adult rats exposed to nicotine as adolescents find alcohol less aversive than adults exposed to alcohol alone.
Our group has recently extended these analyses to assess the effects of adolescent drug history on drug reward and aversion in adulthood. Adolescent rats are interesting in that compared to adults, they are generally more sensitive to a drug’s rewarding effects and less sensitive to their aversive effects, making them much more likely to sample and use such drugs. Given the prevalence of adolescent drug use and the plasticity of the developing brain, we have asked how an adolescent history with nicotine affects alcohol use. Using the taste aversion design to assess the drug’s aversive effects, we have reported that adult rats exposed to nicotine as adolescents find alcohol less aversive than adults exposed to alcohol alone, an effect that can shift the reward/aversion balance of alcohol and increase its use and abuse.
Drug history is but one factor that may impact the relative balance of the drug’s rewarding and aversive effects, and understanding these various factors and how they may affect this balance may be important to predicting abuse vulnerability. This work from our laboratory and elsewhere points to a use of the taste avoidance design not only for characterizing the nature of aversion learning but also as a tool in investigating the basis for other behavior, including drug use and abuse.
Click "American Scientist" to access home page
American Scientist Comments and Discussion
To discuss our articles or comment on them, please share them and tag American Scientist on social media platforms. Here are links to our profiles on Twitter, Facebook, and LinkedIn.
If we re-share your post, we will moderate comments/discussion following our comments policy.