Tardigrades
By William Randolph Miller
These ambling, eight-legged microscopic “bears of the moss” are cute, ubiquitous, all but indestructible and a model organism for education
These ambling, eight-legged microscopic “bears of the moss” are cute, ubiquitous, all but indestructible and a model organism for education

DOI: 10.1511/2011.92.384
The young woman in my office doorway is inquiring about the summer internship I am offering.
What’s a tardigrade? she asks.
Tardigrades, I reply, are microscopic, aquatic animals found just about everywhere on Earth.

Eye of Science/Photo Researchers
Terrestrial species live in the interior dampness of moss, lichen, leaf litter and soil; other species are found in fresh or salt water. They are commonly known as water bears, a name derived from their resemblance to eight-legged pandas. Some call them moss piglets and they have also been compared to pygmy rhinoceroses and armadillos. On seeing them, most people say tardigrades are the cutest invertebrate.
At one time water bears were candidates to be the main model organism for studies of development. That role is now held most prominently by the roundworm Caenorhabditis elegans, the object of study for the many distinguished researchers following in the trail opened by Nobel Prize laureate Sydney Brenner, who began working on C. elegans in 1974. Water bears offer the same virtues that have made C. elegans so valuable for developmental studies: physiological simplicity, a fast breeding cycle and a precise, highly patterned development plan. Some species may, like C. elegans, be eutelic, meaning that the organisms retain the same number of cells through their development. Tardigrades have somewhere over 1,000 cells. I and others use water bears as a model educational organism to teach a wide range of principles in life science.
Tardigrades are nearly translucent and they average about half a millimeter (500 micrometers) in length, about the size of the period at the end of this sentence. In the right light you can actually see them with the naked eye. But researchers who work with tardigrades see them as they appear through a dissecting microscope of 20- to 30-power magnification—as charismatic miniature animals.
Most tiny invertebrates dart about frantically. Tardigrades move slowly as they clamber around on bits of debris. They were first named tardigrada in Italian from the Latin meaning “slow walker.” Tardigrades walk on short, stubby legs located under their bodies, not sticking out to the sides. These stout legs propel them unhurriedly and deliberately about their habitat.
Tardigrades have five body sections, a well-defined head and four body segments, each of which has a pair of legs fitted with claws. The claws vary in different species from familiarly bearlike to strangely medieval fistfuls of hooked weaponry. The hindmost legs are attached backwards, in a configuration unlike that of any other animal. These legs are used for grasping and slow-motion acrobatics rather than for walking.
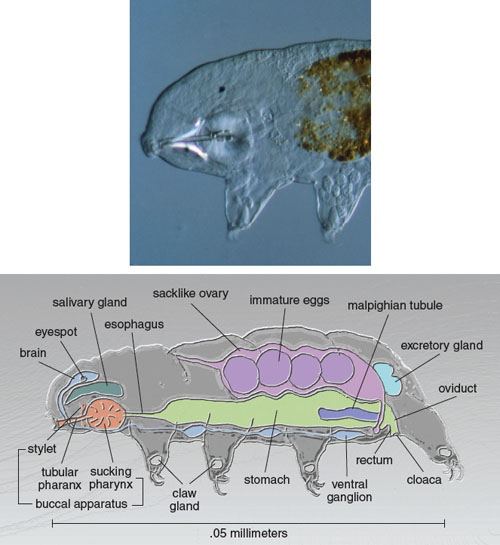
Dr. David J. Patterson/Photo Researchers. Illustration at bottom by Tom Dunne, adapted from a figure by the author.
Inside these tiny beasts we find anatomy and physiology similar to that of larger animals, including a full alimentary canal and digestive system. Mouth parts and a sucking pharynx lead to an esophagus, stomach, intestine and anus. There are well-developed muscles but only a single gonad. Tardigrades have a dorsal brain atop a paired ventral nervous system. (Humans have a dorsal brain and a single dorsal nervous system.) The body cavity of tardigrades is an open hemocoel that touches every cell, allowing efficient nutrition and gas exchange with no need for circulatory or respiratory systems.
Taxonomists divide life on Earth into three domains: Bacteria, Archaea (an ancient line of bacterialike cells without nuclei that are likely closer in evolutionary terms to organisms with nucleated cells than to bacteria), and Eukarya. Eukarya is divided into four kingdoms: Protista, Plantae, Fungi and Animalia. Phylum Tardigrada is one of the 36 phyla (roughly, depending on whom one asks) within Animalia—making water bears a significantly distinctive branch on the tree of life.
Tardigrades are encased in a rugged but flexible cuticle that must be shed as the organism grows. Thus they have been placed among the phyla on the ecdysozoa line of evolution between animals such as nematodes and arthropods that also shed their cuticles to grow.
I can see in my drop-in student’s eyes that she remembers the ecdysozoan way of being from introductory biology but she does not really understand it.
Animals grow in either of two ways, by adding more cells or by making each cell larger. Tardigrades generally do the latter. If an animal has a hard cuticle or exoskeleton, it must break out of that shell in order to grow. For example, in summer in many parts of the world, one encounters the shed exoskeletons of locusts on trees everywhere.
Tardigrades are divided into two classes, Eutardigrada and Heterotardigrada. As a general rule, the members of Eutardigrada have a naked or smooth cuticle without plates, whereas the Heterotardigrada boast a cuticle armored with plates.
Tardigrades’ best-known feature is their brute, dogged ability to survive spectacularly extreme conditions. A few years ago, the Discovery network show Animal Planet aired a countdown story about the most rugged creatures on Earth. Tardigrades were crowned the “Most Extreme” survivor, topping penguins in the Antarctic cold, camels in the dry oven of the desert, tube worms in the abyss and even the legendarily persistent cockroach.
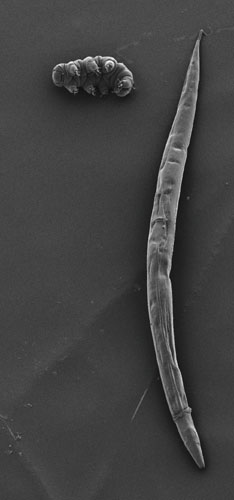
Photograph courtesy of Bob Goldstein and Vicky Madden of the University of North Carolina at Chapel Hill.
But extreme survivorship applies only to some species of terrestrial tardigrades. Marine and aquatic tardigrades did not evolve these characteristics because their environments are stable. It appears that the extravagant survival adaptations have been selected in direct response to rapidly changing terrestrial microenvironments of damp flora subject to rapid drying and extreme weather.
Terrestrial tardigrades have three basic states of being: active, anoxybiosis and cryptobiosis. In the active state, they eat, grow, fight, reproduce, move and enact the normal routines of life. Anoxybiosis occurs in response to low oxygen. Tardigrades are quite sensitive to oxygen tension. Prolonged asphyxia results in failure of the osmoregulatory controls that regulate body water, causing the tardigrade to puff up like the Michelin Man and float around for a few days until its habitat dries out and it can resume active life.
Cryptobiosis is a reversible ametabolic state—the suspension of metabolism—that has inevitably been compared to death and resurrection. In cryptobiosis, brought on by extreme desiccation, metabolic activity is paralyzed due to the absence of liquid water. Terrestrial water bears are only limnoterrestrial—aquatic animals living within a film of water found in their terrestrial habitats. Moss and lichens provide spongelike habitats featuring a myriad of small pockets of water and, like sponges, these habitats dry out slowly. As its surroundings lose water, the tardigrade desiccates with them. It has no choice. The creature loses up to 97 percent of its body moisture and shrivels into a structure about one-third its original size, called a tun. In this state, a form of cryptobiosis called anhydrobiosis—meaning life without water—the animal can survive just about anything.
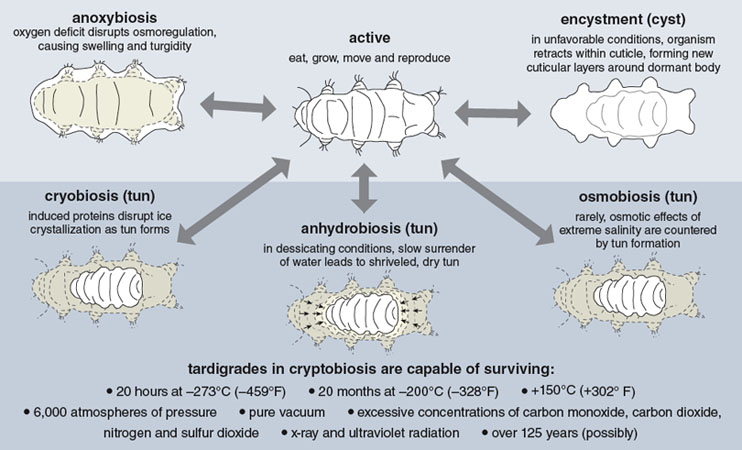
Illustration by Tom Dunne.
Tardigrades have been experimentally subjected to temperatures of 0.05 kelvins (–272.95 degrees Celsius or functional absolute zero) for 20 hours, then warmed, rehydrated and returned to active life. They have been stored at –200 degrees Celsius for 20 months and have survived. They have been exposed to 150 Celsius, far above the boiling point of water, and have been revived. They have been subjected to more than 40,000 kilopascals of pressure and excess concentrations of suffocating gasses (carbon monoxide, carbon dioxide, nitrogen, sulfur dioxide), and still they returned to active life. In the cryptobiotic state, the animals even survived the burning ultraviolet radiation of space.
Challenging student scientists to ponder the astonishing durability of tardigrades brings their understanding of physics, chemistry and biology into play. They recall that water expands as it approaches the freezing point, which is why ice floats. At 4 degrees celsius the expansion of water exerts sufficient force to split boulders, rupture metal containers and explode living cells. A cell is more than 95 percent water. The rupturing forces and icy microshards that form in frozen cells are the same that cause frost bite.
How can water bears survive all that? my new student and, perhaps, future colleague asks.
Really, deep down, we’re still puzzled about a lot of it, I respond.
The survival attributes of tardigrades are in fact quite appropriate for an organism that makes its home in mosses and lichens (bryophytes), which provide them with just a thin layer of protection. Bryophytes are subject to the environmental extremes experienced on a planet bathed in solar radiation. They may receive varying periods of direct ultraviolet exposure and are never far from drying out as ambient conditions change.
Tardigrades exhibit distinctly different responses, grouped under the general name of cryptobiosis, to different sources of stress. Anhydrobiosis and cryobiosis lead to the formation of tuns, but they are not equivalent—they are different mechanisms for protection against different environmental assaults.
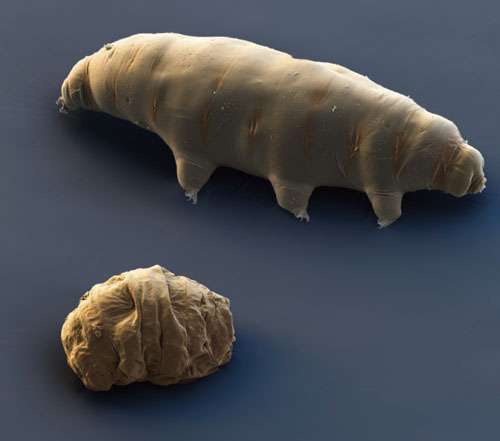
Eye of Science/Photo Researchers
Anhydrobiosis—metabolic suspension brought on by nearly complete desiccation—is a common state for tardigrades, which they may enter several times a year. To survive the transition, water bears must dry out very slowly. The tun forms as the animal retracts its legs and head and curls into a ball, which minimizes surface area. When nearly all of its internal water has been surrendered, the tardigrade is in anabiosis, a dry state of suspended animation. It is almost as if the animal preserves itself by becoming a powder comprised of the ingredients of life. When rehydrated by dew, rain or melting snow, tardigrades can return to their active state in a few minutes to a few hours.
In cryobiosis, another form of cryptobiosis, the animal undergoes freezing yet can be revived. Any temperature below the cell cytoplasm’s freezing point suppresses molecular mobility and therefore suspends metabolism. Deep-freeze temperatures could be expected to cause additional structural disruptions, yet tardigrades, as noted above, have survived the most drastic chills. It seems likely that survival is conferred by the release or synthesis of cryoprotectants. These agents may manipulate tissue freezing temperature, slowing the process and allowing an orderly transition into cryobiosis, and they may suppress the nucleation of ice crystals, resulting in an ice-crystal form that is favorable for subsequent revival with thawing.
Osmobiosis is a response to extreme salinity, which can cause destructive osmotic swelling. Some tardigrades exhibit strikingly effective osmoregulation, maintaining stasis in the face of steep osmotic gradients. Some others escape via formation of a tun that is impervious to osmotic transfer.
In 2007, tardigrades became the first multicellular animal to survive exposure to the lethal environs of outer space. Researchers in Europe launched an experiment on the European Space Agency’s BIOPAN 6/Foton-M3 mission that exposed cryptobiotic tardigrades directly to solar radiation, heat and the vacuum of space. While the experimental vessel orbited 260 kilometers above the Earth, the researchers triggered the opening of a container with tardigrade tuns inside and exposed them to the Sun. When the tuns were returned to Earth and rehydrated, the animals moved, ate, grew, shed and reproduced. They had survived. In summer of 2011, Project Biokis, sponsored by the Italian Space Agency, ferried tardigrades into space on the U.S. space shuttle Endeavor. Colonies of tardigrades were exposed to different levels of ionizing radiation. The damage is now being assayed to learn more about how cells react to radiation and, perhaps, how tardigrade cells fend off its damage.
My student’s mind is turning.
How far is the nearest solar system? Could cryptobiotic tardigrades make it to Earth? she asks.
I saw a post recently on a listserv that says it might be only about 10 light years away, I answer. So if tardigrade tuns were transported here on a meteor or asteroid at just one tenth the speed of light, they could conceivably make the trip within the known survival capability of the animal. Theoretically. But the likelihood is awfully small. And think of the poignancy of arriving after that great journey but having no way to make it through the fiery descent through an atmosphere. Even tardigrades can’t survive that.
Surviving intense radiation suggests an especially effective DNA repair system in an active organism. Effective osmoregulation in extreme salinity implies a vigorous metabolism—osmoregulation in the face of high environmental salinity is energetically extremely expensive as metabolic transactions go, requiring the pumping of ions against steep osmotic and ionic gradients. Thus, we see in tardigrades two opposing responses to environmental extremes: the passive response of dormancy in the form of cryptobiosis, balanced by the hyperactive responses of impressive DNA repair and high-performance osmoregulation. As practitioners of adaptive evolution, tardigrades are virtuosos.
Tardigrades have been discovered just about everywhere that anyone has looked, from the Arctic to the equator, from intertidal zones to the deep ocean, and even at the top of forest canopies. Their ubiquity is intimately linked to their survivorship. I am often asked how tardigrades manage to find their way to the canopy of towering trees. Most likely, wind carries them. In the tun state they are barely distinguishable from dust particles. But like spores, pollen and seeds, the tuns have a preference for where they land. Many microenvironments will be unsuitable habitats for freshly arrived tardigrades. Yet an unhappily placed tun can simply wait for a change in precipitation or perhaps a change in season. When conditions improve, life can begin again.
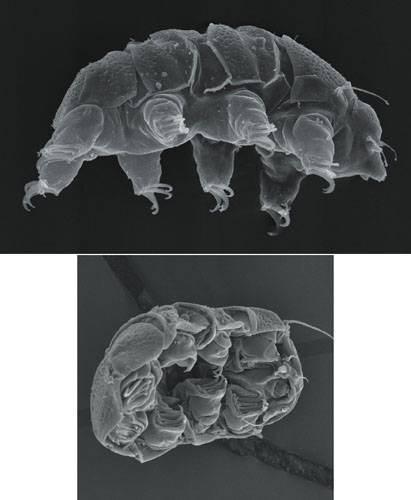
Images courtesy of the author.
Contributing to their success as travelers is the fact that many tardigrades of moss, lichen and leaf litter are parthenogenetic, able to produce eggs without mating, and in a few cases are hermaphroditic, able to self-fertilize. A lone tardigrade on an ill wind—active, tun or egg—may be able to establish a population where it lands if the habitat is suitable. We may be under tardigrade rain right now.
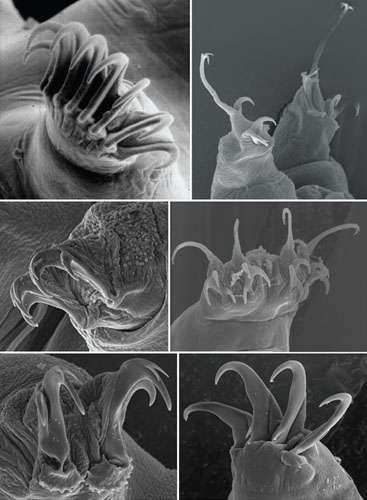
Images courtesy of the author and Clark W. Beasley of McMurry University.
At present there are about 1,100 described species of water bears, but not all are valid. Some descriptions are repeats and some are just plain flawed. Around 1,000 species have been properly identified and described. We have about 300 marine, 100 freshwater and 600 terrestrial species. But the land species are much easier to find and have been pursued by many more researchers over many more years. Still, my students have discovered and described four new species so far, and we are working to confirm another half dozen, including one found on the campus of Baker University in Kansas, where I am a faculty member. We believe there is an abundance of species yet to be discovered, especially in the nonterrestrial environments.
I might discover a new species? she asks.
Yes, sitting at a microscope, you might observe an animal nobody in the world has ever seen before. That is pure exploration. In the blink of an eye, you might find a clue to the evolution of the phylum or identify the animal that holds the cure for cancer, I say. Then again, you might not. It took me 16 years to find my first new species.
Last summer, the student who inquired at my office, Rachael Schulte, became an intern working on our National Science Foundation grant under the Research at Undergraduate Institutions (RUI) program designed to teach research by exploring and expanding the biodiversity of the phylum Tardigrada in North America.
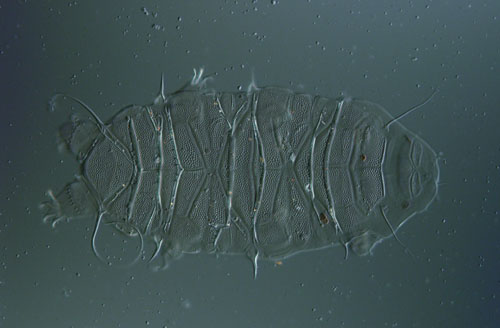
Image courtesy of the author and Rachael Schulte.
After a couple of weeks of practice on lichen from local trees, Rachael had become proficient with the tools of the tardigrade trade—the dissecting scope, the wire Irwin loop, slide preparation, imaging, record keeping and identification to the level of genus. She was ready to work on actual research material, so we set her up with samples collected a couple of years before on a transect from more than 9,000 feet up in the Sierra Nevada Mountains down to Fresno, California.
Just a week later, she came to me with a finely made slide.
I think it is a Pseudechinsicus but it has many small, plates across its back, lateral filaments on the edge of each segment, and a toothed collar on the last legs, she says.
This was a teaching and a learning moment. I put the slide on the stage of our computer-imaging microscope to take a look.
Do you have other specimens?
Eight, all from the same sample, she replies.
I have seen this before, it is Pseudechinsicus because of this pseudo, or false, segmental plate. But it appears to also have a pseudosegmental plate on each segment. Let’s see how it keys out in Ramazzotti and Maucci, I say.
In 1983, Giuseppe Ramazzotti and Walter Maucci published the monograph The Phylum Tardigrada. It was translated from Italian into English by Clark Beasley in 1985. It is now 27 years out of date and includes only half of the described species. But it remains the reference of first resort. We started with the genus Pseudechinsicus. As I read the diagnostic questions in the key, Rachel worked the microscope to answer them.
The animal looked like Pseudechinsicus raneyi, as described by Gragrick, Michelic, and Schuster in 1964. We pulled up a copy of the paper from the files (we have PDF files of 95 percent of all tardigrade papers) and read. The description matched our animal. We then looked at the 1994 list of species, along with the relevant research papers and geographic distributions, prepared by McInnes. There were only two listings for our species—the original description from California and Schuster and Gragrick’s entry in their 1965 classic work on the western North American tardigrades, which added Oregon to Pseudechinsicus raneyi’s known range. Searching through the more recent literature in our database, Rachael discovered that I had also found the creature in Montana during my master’s work at the University of Montana, Missoula, in the late 1960s, although I did not publish the record until 2006. Now after 40 years we had a fourth record and a new location for an uncommon regional animal.
During our literature review, we learned that the genus was described by Gustav Thulin in 1911, who gave high taxonomic value to the presence of the pseudosegmental plate. Then in 1987, Kristensen revised the family Echinsicidae, redescribed the existing genera and added four new ones to the list. Because this occurred after our creature was described, we needed to confirm the genus assignment by reviewing its characteristics against the amended, more detailed description.
We started down the list of characteristics under the genus Pseudechiniscus I read the first line:
Echiniscidae with black eyes: rigid buccal canal, stylet supports may be present, but very tiny and located close to the margin of the pharyngeal bulb.
Looking through the microscope, under oil immersion at 1,000-times magnification, Rachael says:
Black eyes, yes, but the buccal canal is long and bent. Flexible. No visible stylets.
I continue, … unpaired scapular plate. Typical tiny basal secondary spurs.
No, it is paired. And the spurs are not tiny, nor basal, she says.
I look up and say, No mention of the extra pseudosegmental plate.
Our specimens did not match the description of the genus Pseudechinicus. So we checked the other generic descriptions within the family the same way and concluded that our specimens matched none of them. We now thought there were enough significant deviations from the existing descriptions to merit describing and naming a new genus.
Over the next several months we borrowed the original type specimen of Pseudechiniscus raneyi from the Bohart Museum at the University of California at Davis and confirmed that it was the same as our specimens. Rachael and I made images of the slides, measured multiple characteristics on each specimen and developed a comparative table. We checked and double checked our specimens. As we started to pass the draft of a manuscript back and forth, I asked Rachael whether she wanted to be a coauthor describing the new animal or to have it named after her.
I get to choose?
You found it, helped detect the differences, and contributed to the new description, I say.
What is the difference? she asks.
Both are a bit of immortality. But you can’t be an author and namee, I say. Normally, we would name a new genus after some unique characteristic of the organism, or to commemorate a fellow researcher. We could name the genus after you, but the species would remain raneyi because our specimens match the already described Pseudechiniscus raneyi. We are simply moving an existing species into a new genus, so the species name does not change, I explain. But if you are an author of the paper describing the new genus, your name goes after the genus name, before the date, every time the genus is listed, I say.
So what do we name it? she asks.
What is the most distinctive feature of the critter?
The elongated buccal tube? she says.
True, but that is very difficult to see. What else have you seen?
The extra pseudosegmental plates? she suggests.
Okay, how about Multi pseudechiniscus? I ask.
Rachael presented a poster about the discovery at the November 2010 Sigma Xi International Meeting and Student Research Conference in Raleigh, North Carolina, with 250 other undergraduate researchers. The new genus of water bear is shown in Figure 8. Our manuscript reporting the find is under review at a peer-reviewed journal.
Click "American Scientist" to access home page
American Scientist Comments and Discussion
To discuss our articles or comment on them, please share them and tag American Scientist on social media platforms. Here are links to our profiles on Twitter, Facebook, and LinkedIn.
If we re-share your post, we will moderate comments/discussion following our comments policy.