Risks and Benefits of Radiation
By Timothy J. Jorgensen
The story of radon's study in public health can be a guide for how to best weigh the pros and cons of radiation use.
The story of radon's study in public health can be a guide for how to best weigh the pros and cons of radiation use.

On December 2, 1984, Stanley J. Watras, an engineer working on construction of the new Limerick nuclear power plant near Pottstown, Pennsylvania, arrived at work.
The plant was scheduled to begin generating power within three weeks, and the construction crew had just installed radiation detectors at the plant doors—a standard safeguard to ensure that nuclear workers don’t exit the plant with any radioactive contamination on their bodies. When Watras arrived that day, he set off the alarms on the detectors as he walked into the plant. Over the following two weeks he would set off the alarms every morning. Further investigation revealed that his clothes were contaminated with radioactivity that he had picked up at his home!
Image courtesy of Hyo-eun (Carrie) Bhang and Martin Pomper, and the National Cancer Institute.
When radiation safety personnel from the plant visited Watras’s home, they discovered what they didn’t think possible. There was more radon gas in the Watras house than was found in a typical uranium mine—nearly 20 times as much! Surprised, the radiation safety technicians checked the radon levels in the neighboring houses. “Our house,” Watras remarked in consternation, “had perhaps the highest contamination level in the world, but our next door neighbors had none.” How could this be?
The Watras house was located on the Reading Prong, a geological formation full of uranium deposits in the eastern United States. Radon, a radioactive gas produced through the long decay chain of uranium into other isotopes, leaks from the ground in this area and then mixes with aboveground air. The fact that radon seeps from the ground over uranium- containing earth had been known since 1908. Nevertheless, geologists hadn’t previously appreciated how spotty radon leakage could be. Some ground locations can have virtually no leakage, while a spot a few hundred feet away could have huge amounts of radon streaming out.
Just as water travels great distances along crevices in bedrock, subterranean radon travels along ground faults to emerge as a "spring" of gas.
As it turns out, subterranean radon behaves like subterranean water. Just as underground water gathers in pockets and travels great distances along crevices in the bedrock, often to emerge in discrete locations on the ground surface in the form of a natural spring of water, radon travels along ground faults to emerge as a “spring” of gas. The Watras home was built right on top of a spring of radon gas. How many other houses built on radon springs were out there and, more importantly, how much danger did they pose to their residents?
The Watras family had moved into their house in January of that year, so they had been exposed to the radon for less than a full year. Nevertheless, doctors told them, based on risk estimates of the U.S. Environmental Protection Agency (EPA), their brief exposure to radon made them seven times more likely to die of lung cancer within 10 years compared to a person without radon exposure. Their three young children might not make it to adulthood.

The Morning Call
The family moved out of the house immediately and tried to resume their normal lives. As Stanley Watras explained at the time, “[If we] keep worrying about it, we might not live long enough to see whether the doctors were right, because depression and psychological pressure would be too much for us to survive.”
It had been firmly established since 1944 that breathing a lot of radon carries a substantial lung cancer risk. But after that, things get a little murky. Why? Because another major cause of lung cancer poses a much bigger threat than radon, and this of course is cigarette smoking. Cigarette smoke produces a statistical haze through which all radon data must be viewed.
Expert panels have evaluated mountains of data on miners in an attempt to precisely determine the amount of lung cancer risk that could be attributed specifically to radon exposure, rather than to smoking, but their analytical efforts were hampered. This is not only because smoking was so prevalent among miners and nonminers alike, but also because smoking makes the lung cancer risk from radon worse—much worse.
It’s not clear why smokers are hypersensitive to radon. It could be because radon and the chemicals in cigarette smoke damage DNA in somewhat different ways. For example, the ionizing radiation from radon tends to break DNA, whereas the highly reactive chemicals in cigarette smoke tend to attach themselves to the DNA and form what chemists call bulky adducts (because they represent large chemical additions to the DNA structure). These different forms of DNA damage may impede each other’s repair. Thus, having both types of DNA lesions at the same time might be much worse that having one type alone.
Alternatively, smokers may be hypersensitive to radon because they have damaged their lungs to the point that their bronchi—the treelike tubes of air passageways—are no longer able to efficiently remove particles that are breathed in. Particles laden with radon may stay in the smoker’s lungs longer, resulting in a higher radiation dose to a smoker’s lungs than to a nonsmoker’s lungs, even when they’re breathing the same amount of radon. It’s not clear which of these two possible mechanisms is correct. It might even be both, or perhaps neither. Nonetheless, smokers definitely are at higher risk of lung cancer caused by radon—six to eight times higher.
In order to set limits to radiation dose, you need to be able to measure it. A dose estimate strategy for radon is to make use of a simple mathematical relationship known as Haber’s rule for gaseous hazards, D = C x E, where D is dose, C is concentration, and E is exposure time. Haber’s rule tells us that dose is approximately proportional to both the concentration of the noxious agent in the air and the duration of exposure to that agent. More scholarly dosimetric models may give more precise dose estimates, but Haber’s rule seems to be remarkably accurate in predicting health risks from gases, without any of the complicated statistical hoopla that surrounds more erudite dose modeling.

Photograph courtesy of Nathaniel Brooks, the New York Times, and Redux Pictures.
Radon is one of the few radiation hazards not typically measured in millisieverts (mSv), the standard international unit for radiation dose equivalence. Instead, Haber’s rule has been invoked to provide a unit unique to radon. Since most of the data comes from mine workers, the experts have chosen to calculate lung cancer risk from radon using a unit called the working level month (WLM). Although it has a technical and precise definition, for our purposes it can just be defined as the radon dose that a typical miner might receive from working in a typical radon-contaminated mine for one month. Simply stated, it is just the radon concentration of the mine, expressed in working levels (WL), multiplied by the exposure time measured in months (M), à la Haber’s rule.
Expressing radon dose in WLMs also has another practical advantage. It allows us to depict other radon exposures, such as living in a radon-contaminated home, in terms of the equivalent amount of mining work it would represent.
Because “the dose makes the poison” (a famous quote by Swiss Renaissance physician Paracelsus [1493–1541]), we need dose-response data to identify the dose where radon levels become a health concern. Because lung cancer is the health effect of interest, “doses” (i.e., WLMs) are typically plotted against lung cancer incidence rates, and fitted to a straight line. The fitted line is drawn straight, rather than curved, because epidemiologists assume cancer risk is directly proportional to radiation dose at all dose levels, no matter how low those doses are. Most scientists believe that there is a finite probability of contracting cancer from any exposure to a carcinogen, because there is no dose so low that DNA damage does not occur, and because DNA damage is the known precursor to cancer. Epidemiologists call this the linear no threshold (LNT) model of cancer risk assessment.
Not all scientists favor linear models. Some strongly believe that the LNT approach to radiation protection has exaggerated the risk of cancer from low radiation doses, and their arguments have some merit. In contrast, no credible scientists believe that the LNT model underestimates low-dose risk. Thus, the LNT model is generally considered the most conservative, affording the greatest degree of protection to the public.
We’ve taken the long route to get to the final radon risk estimates, but understanding the methods and approaches used in risk assessment does three things: It allows us to appreciate the strengths of the risk estimates, to recognize their weaknesses, and to gauge our level of confidence in their accuracy. Now that we know how radon estimates are generated, let’s take a look at the final numbers.
The scientific experts’ best estimate of the lung cancer risk per WLM is approximately 0.097 percent for smokers and 0.017 percent for nonsmokers. Stated simply, working for one month in a typical uranium mine increases the odds of getting lung cancer by about 1 in 1,000 for a smoking miner, and 1 in 6,000 for a nonsmoking miner.
Another way of expressing the risk of one month of work is as follows: For every 10,000 smoking miners that worked in a mine for one month, we would expect 10 of them to eventually contract cancer from their radon exposure. Alternatively, if 10,000 nonsmoking miners worked for one month, we would expect fewer than two of them (i.e., 1.7 miners) to get lung cancer.
Image courtesy of the Pennsylvania Department of Conservation and Natural Resources.
All right you say, so much for the mines. What about my home? Here’s the EPA’s logic on home radon risks. The EPA has decided that a home resident’s radon dose should be no more than 2 percent of that of a typical mineworker. That means the concentration of radon in home air should be at or below 0.02 WL. To go much lower than 0.02 WL would be technically difficult (i.e., extremely expensive) and practically unwarranted, given that even outside air has an average radon concentration of 0.0025 WL. What is the risk level to residents living their entire life in a home with radon concentrations at 0.02 WL? According to the EPA’s calculations, if a person lived in that home every day of his life, occupied the house for up to 17 hours per day (70 percent of the time), and lived to be 75 years old, then that person’s lifetime risk of lung cancer from the radon in his house would be 6.2 percent if he were a smoker, but only 0.73 percent, if he were a nonsmoker. But exactly how bad are these levels of risk? Let’s explore this question.
Image courtesy of the EPA.
Instead of starting with 10,000 people and tracking their health outcomes as we did above for the miners, another way to portray risk is simply to ask the following question: “How many people would need to receive this particular dose level before one of them would be expected to have a harmful outcome (e.g., lung cancer)?” In other words, what number of people need to be exposed in order to harm just one? When risk estimates are framed around this question, the risk metric is called the number needed to harm (NNH).
Restating the lung cancer risks from lifetime residence in a home at the EPA radon limit in terms of NNH units yields the values of 16 and 137 for smokers and nonsmokers, respectively. This means that out of 16 smoking lifelong residents of such radon-containing houses, 1 would be expected to develop lung cancer from the exposure, while it would take as many as 137 nonsmoking lifelong residents for 1 to become afflicted with lung cancer.
If these radon risk levels still seem high to you, consider this. The chance of a nonsmoker contracting a fatal cancer of any type during her lifetime is approximately 25 percent, a number that is relatively high because cancer is an unfortunately common disease. The additional risk from living in the house with radon at the residential limit, according to the numbers above, would increase that risk from 25 to 25.73 percent. But smokers, in contrast, already have an elevated baseline risk of developing a fatal cancer approaching 50 percent (for a heavy smoker), apart from any radon exposure they might have. For a smoker, the risk of developing a fatal cancer during a lifetime thus moves from about 50 to 56.2 percent.
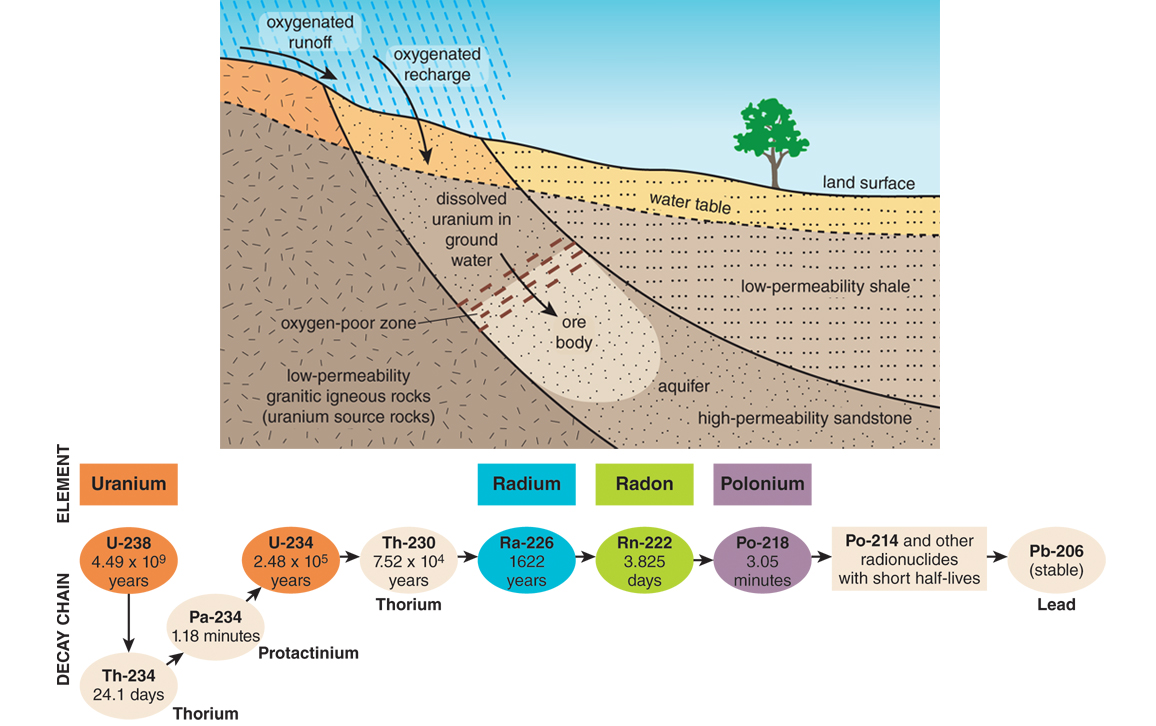
Illustrations by Barbara Aulicino.
Also, consider the fact that virtually no one matches the EPA’s extreme assumptions about the length of home residency. Thus, the actual doses to residents living in houses at the EPA exposure limit of 0.02 WL are probably less than a tenth of the theoretical worst-case doses and, likewise, their lung cancer risk would also be less than a tenth of the risk levels calculated above.
Based on the EPA’s risk assessment numbers and their underlying assumptions, the agency has claimed that radon in homes theoretically kills as many as 21,000 Americans every year. This amounts to 13 percent of the 160,000 annual lung cancer deaths in the United States, and 3.5 percent of the 585,000 total annual cancer deaths of all types. But who are these “theoretically” dead people anyway?
Remember that smokers are six to eight times as likely to get radon- induced lung cancer, and consider that the prevalence of smokers in the U.S. population is about 18 percent. Even if you accept the EPA’s estimate that 21,000 annual lung cancer deaths in the United States are due to radon exposure in homes, about 19,000 of those dead would be smokers. Only 2,000 deaths would occur among nonsmokers, just 0.3 percent of the total annual cancer deaths and about one-tenth of the number of annual deaths from influenza. In short, you must be very unlucky indeed to die from radon-induced lung cancer if you’re not a smoker.
When the EPA first issued its radon guidelines in the 1980s, the public was slow to comply. Social psychologists say that is likely because radon is a natural hazard, as opposed to a manmade hazard. For whatever reason, people seem to be less afraid of natural hazards than of manmade ones. So the EPA increased its hype with an advertising campaign that focused on the extreme situations and downplayed the fact that the lung cancer risk of radon was largely restricted to smokers.
The public was slow to comply with EPA radon guidelines in the 1980s, so the agency increased its hype with a campaign that downplayed the fact that the cancer risk was largely restricted to smokers.
The EPA’s well-publicized claim that radon is the second leading known cause of lung cancer, after smoking-related causes, was also misleading. Although the statement is strictly true, it implies that the leading cause of lung cancer among nonsmokers is radon exposure. This interpretation is false. Radon is estimated to account for 26 percent of lung cancers among nonsmokers. The largest proportion of lung cancers among nonsmokers is actually due to other causes that are currently unknown (40 percent). So, lung cancer among nonsmokers is rare, and radon-induced lung cancer among nonsmokers is rarer still.
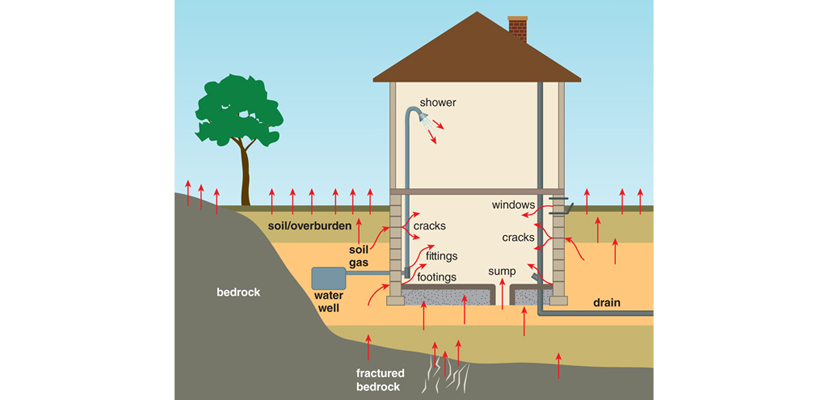
Illustration by Barbara Aulicino.
Many scientists and risk assessors felt that the EPA had twisted the facts to advance its own agenda. This resulted in some pretty ugly fighting between the agency and the scientific community. The controversy spilled over into the public arena and led many people to the erroneous conclusion that the risks from home radon were not real. They are real. But the level of the radon risk to homeowners was certainly overstated by the EPA, at least in the early years.
Thirty years later, the rhetoric has died down. A visit to the EPA’s public radon website today reveals that it is doing a better job of accurately characterizing the risk of radon, and it is more candid about the fact that radon is primarily a problem for smokers. The EPA has not, however, relaxed its level for radon in homes.
The Watras family members were probably the main victims of the EPA’s miscommunication of risk. They were led to believe that their lives were in serious peril, and they carried that psychological stress for decades. The truth is that they suffered a moderate increase in cancer risk from their radon exposure, which was hardly a death sentence.
Whatever happened to the Watras family? Once the family moved out of the house, the EPA used it as a testing laboratory for various radon remediation technologies. After many months, the agency was able to lower the radon levels to the recommended limit, and the Watras family moved back in. Stanley and his wife Diane are still living in the house (as of 2015). Their children are now grown. After 30 years, none of them has died of lung cancer, although the EPA risk estimates suggested that nearly all of them would have done so by now. How could the dire predictions for the Watrases have been so off the mark?

Photograph courtesy of the National Cancer Institute.
This is one of the trade-offs of using multiple, highly conservative assumptions in risk assessment. It may seem prudent to inflate the risk in order not to underestimate it. Nevertheless, by adopting high-end estimates for every uncertain risk parameter, the cascade of high-end risk assumptions can compound to the point where the final predicted risk levels become incredulous and may even defy common sense. For example, according to the EPA’s risk estimates, just one year of living in the Watras house (16 WL) would put a smoker’s lifetime lung cancer risk from radon exposure at 56 percent, in addition to his lung cancer risk from the smoking (15 to 50 percent, depending upon his smoking level), making his overall lung cancer risk as high as 100 percent. But as Naomi Harley, a professor in the Department of Environmental Medicine at New York University, has pointed out, the lung cancer death rates for miners with the highest radon exposures ever recorded (nearly all of whom were smokers) were never greater than 50 percent. Thus, estimates of lung cancer risk from radon in homes that exceed 50 percent must be considered suspect.

The Morning Call
There is absolutely no benefit from living in a radon-contaminated house, so it might seem warranted to completely eliminate radon as a source of radiation exposure. But what about diagnostic radiography, for which the benefits to health are potentially enormous? Is it wise to forgo diagnostic x-rays, simply because of their cancer risk, and thereby deprive ourselves of their benefits? To address this question, let’s use the example of an x-ray of a broken arm.
Modern x-rays of broken arms give an effective dose of about 0.001 mSv to the patient. The term “effective dose” means that because an arm is about 5 percent of total body weight, the risk associated with an arm dose of 0.020 mSv gives effectively the same fatal cancer risk as a dose of 0.001 mSv to the whole body (i.e., 5 percent of the local dose).
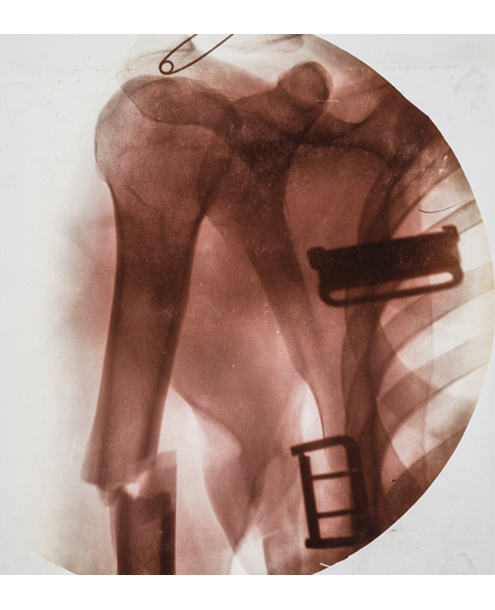
Gustoimages/Science Source
Just as we used lung cancer in mine workers to calculate the risk from radon exposure, we can likewise use studies that were done of atomic bomb survivors to calculate the cancer risk of x-rays. The risk of contracting a fatal cancer from a whole-body radiation exposure, as determined from the atomic bomb survivor studies, is about 0.005 percent per mSv. Therefore, the lifetime risk of contracting cancer from an arm x-ray is 0.001 mSv×0.005 percent, giving a risk estimate of 0.000005 percent (or odds of 1 in 20,000,000). At these odds, you are more likely to win the megalottery than to get cancer from the x-ray exposure of your arm. How many people break their arms in the United States every year? About 1,300,000. Being that the NNH for an x-ray of a broken arm is 20,000,000, whereas only 1,300,000 are actually breaking their arms, it can be seen that we wouldn’t expect any of these people to suffer cancer as a consequence of their arm x-ray. In contrast, virtually all of them will benefit from the medical information that their doctors will glean from the x-ray image of their broken arm. With these numbers, you now have all the information you need to decide whether broken arm x-rays are worth the cancer risk.
Of course, life is more than broken arms. Effective doses have been calculated for virtually every standard diagnostic radiography procedure. In most cases, the risks are highly defined and quantified, but the benefits are less defined and more qualitative. Nevertheless, one thing can be said with confidence: When it comes to using diagnostic radiography for finding the cause of clinical disease symptoms, the benefits almost always far outweigh the risks, largely because the risks are so very low. The only exception to this would be when a doctor orders an unwarranted diagnostic procedure. In that case, the patient receives a risk with no potential for benefit. Not good.
There are many diagnostic radiography procedures out there and we can’t review them all. Nevertheless, all diagnostic radiography procedures fall into one of two categories of use: finding disease in patients who present with clinical symptoms of disease, or screening for disease in people who have no symptoms. For radiographic procedures in the first category, such as the broken arm example above, the benefits nearly always far outweigh the risks. This is largely because the penalty for not finding the underlying cause of the disease can be severe—a nonfunctioning arm or even death—and the cancer risks of partial body radiography procedures are quite low. But for the second category—screening of clinically healthy people for hidden diseases—we should take pause. At this point the jury is still out on the value of these procedures because the issues are much more complicated. For radiographic screening procedures, the cancer risks are often well defined, yet the benefits can be questionable, so medical consumers should proceed with caution.
The ability to convert a radiation dose to a risk estimate levels the playing field for the general public. Armed with an accurate estimate of risk for a whole variety of radiation exposure scenarios, from natural (radon) to manmade (diagnostic x-rays), we now have the tools we need to make decisions regarding which radiation exposures are acceptable and which are not. Most importantly, we now have the means to make an informed decision about controlling our radiation risks. Now it’s up to us to be worthy stewards of this knowledge, and put it to good use for the betterment of our health.
We know much more about the cancer risks of radiation than those of any other carcinogen. So, if we, as reasonable and intelligent people concerned about our health and the health of others, can’t come to a workable consensus about acceptable levels of cancer risk from the use of radiation, there is little hope that we will be able reach a consensus on any other cancer hazards for which very much less is known. On the flip side, however, if we can agree on general principles by which cancer risks of radiation will be addressed, these same principles can likely be co-opted for use in comparable risk situations, in which the data are scarcer, but the need no less compelling. Radiation risk is thus an excellent model for how best to handle environmental cancer threats in general.
This article is excerpted and adapted from Strange Glow: The Story of Radiation by Timothy J. Jorgensen. Reprinted by permission of Princeton University Press.
Click "American Scientist" to access home page
American Scientist Comments and Discussion
To discuss our articles or comment on them, please share them and tag American Scientist on social media platforms. Here are links to our profiles on Twitter, Facebook, and LinkedIn.
If we re-share your post, we will moderate comments/discussion following our comments policy.