Rapid Evolution in Eggs and Sperm
By Katrina G. Claw
Sperm and eggs are ubiquitous and diverse. What drives them to diverge?
Sperm and eggs are ubiquitous and diverse. What drives them to diverge?

DOI: 10.1511/2013.102.210
The experience of learning about the birds and the bees is almost universally awkward, bringing to mind textbook cartoons of the male and female reproductive tracts viewed with amazement, confusion or embarrassment. Although most people do not contemplate reproductive systems every day, speaking of sex and reproduction only in secretive tones, like it or not, we are surrounded by sperm and eggs. On a typical day, you might enjoy a delectable breakfast of fried eggs (a chicken egg and the accompanying yolk) and take a walk outside that triggers a sneeze caused by pollen (plant sperm). You might decide to take a dip in the ocean, where external fertilizers such as sea urchins and abalone are spawning their eggs and sperm throughout the water.
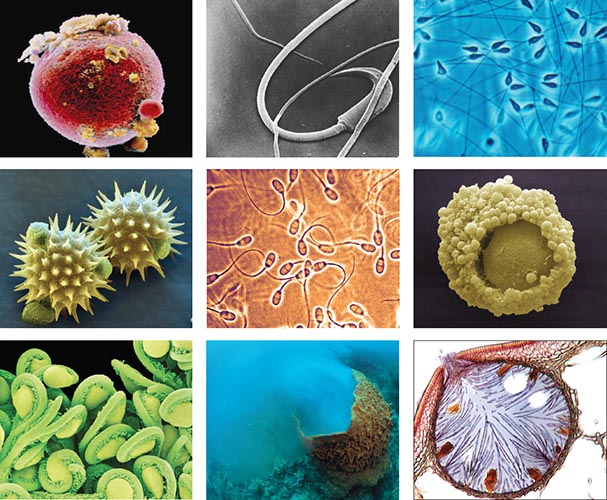
Motta & Familiari, David M. Phillips, Biology Media, David M. Phillips, Dr. Keith Wheeler, Michael Patrick O’Neill, Susumu Nishinaga, Eye of Science, David M. Phillips/Science Source
Sexual reproduction is ubiquitous across plants and animals—more than 90 percent of vertebrates reproduce sexually. Sex is largely responsible for the biological diversity that first fascinated and continues to occupy biologists. Differences in shape, size and coloration, among other characteristics, are often attributed to the evolutionary influences of sex. But why did sex evolve in the first place? Observing sperm and egg diversity across species brings more questions to mind: Why are there more sperm than eggs? How does sexual promiscuity affect the evolution of the sperm and egg? One might think that the same mechanisms, sexual traits and reproductive genes are involved in sperm–egg composition and interactions. But is this the case?
The sperm and the egg vary in looks and structure. Each sperm or egg cell contains exactly half of the genetic information needed to make an individual. In humans, that amounts to 23 chromosomes, one of which is the sex chromosome (X chromosome in eggs and either X or Y chromosome in sperm). The other 22 chromosomes that do not contribute to sex determination are called autosomes. In the common fruit fly, Drosophila melanogaster, sperm and egg cells have three autosomes and an X or Y sex chromosome pair. Unlike in humans, the ratio of X chromosomes to autosomes determines sex in the fruit fly. Figure 1 illustrates the astounding morphological diversity of sperm and egg from various organisms spanning plants, mammals and invertebrates.
An important characteristic that may influence evolutionary dynamics of such diversity is whether an egg is fertilized inside or outside the female’s body. Internal fertilization occurs inside the body and requires some type of insemination by the male, as in many mammals including humans. External fertilization, or free spawning, refers to species in which the sperm and eggs are released into the open environment, as in many marine invertebrates and some plants. Because external fertilization exposes the sperm and egg to a potentially hazardous environment without parental protection, these gametes often develop unique features for their protection and dispersal. For example, because plant pollen is immobile, some plant species have evolved pollen spines for better dispersal and for sticking to fertile flower parts (Figure 1, middle row on far left). Dispersal, or pollination, in plants can occur by animals, wind or water, and the wide variety of pollen morphology reflects the varying pressures to adapt to the surrounding environment.
Sperm and egg also differ dramatically in size and number. Typically, the female invests more into making an egg and will only release a few hundred in her lifetime compared to the ease and abundance of male sperm. Not only does the egg have multiple barriers to avoid polyspermy (multiple sperm entering the egg), it also contains nutritious resources for the new zygote once fertilization has occurred. Because of this, eggs are often larger than sperm. The diameter of a human egg is about the width of a human hair, and a sperm is one-twentieth that size. But sheer numbers make up for what sperm lack in size. A human male will ejaculate about 200 to 500 million sperm. A man ejaculating 20 to 50 million sperm is considered sterile. Human sperm count is modest in the animal kingdom. By contrast, the common farm pig will expel 8 billion sperm in a single emission!

Image on left from T. Burkhead, Promiscuity: An Evolutionary History of Sperm Competition, Harvard University Press, 2002. Image at right Power and Syred/Science Photo Library.
The size and shape of sperm vary between species. One of the biggest sperm is found in the puny common fruit fly. Researchers in Scott Pitnick’s lab at Syracuse University showed that the common fruit fly’s sperm could be measured with a ruler at a whopping two millimeters. But the winner in this sperm size contest goes to a related species of fruit fly, Drosophila bifurca, with a sperm length of 60 millimeters (Figure 2). The D. bifurca sperm is 1,000 times longer than a human sperm. How can such disparate forms of sperm and egg have evolved to perform a well-established function?
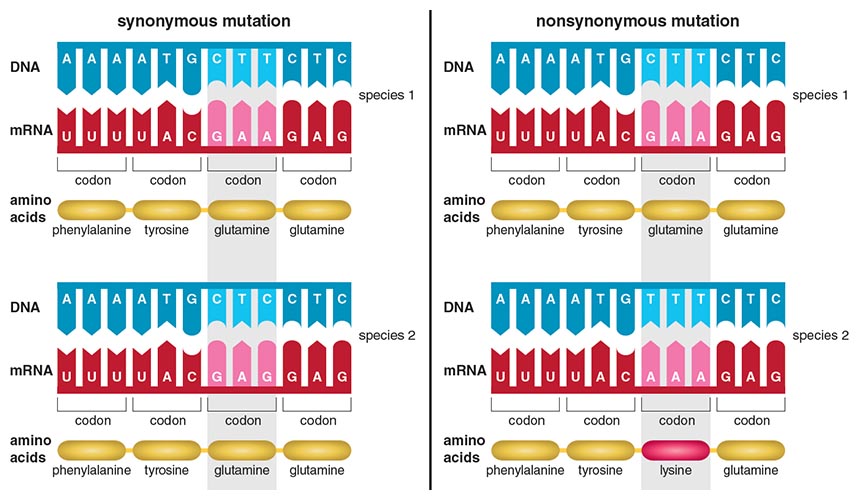
Illustration by Barbara Aulicino.
Various proteins, encoded by genes, make up sperm and eggs. Changes in these proteins over time may result in a single population becoming two separate, distinct species. In 2002, Willie Swanson and Victor Vacquier, respectively of University of Washington and the Scripps Institute of Oceanography, reviewed the many studies that have established that reproductive genes are some of the most rapidly evolving genes in the human genome, along with genes involved in immune response. Proteins are made up of amino acids, and changes in the nucleotide bases that make up genes can change the recipe of amino acids for a protein. Rapidly evolving genes have extremely high rates of amino acid change. As genome sequencing has become more affordable, this general trend is seen in a wide variety of other sequenced species, such as primates, rodents, fruit flies and butterflies. Swanson’s lab, which I joined in 2009, has shown that genes encoding for proteins found on the surfaces of the sperm and egg (potentially involved in sperm–egg interactions) are rapidly evolving in the marine invertebrate abalone (of the genus Haliotis). Our lab also found an important protein involved in forming the egg coat in primates to be rapidly evolving, and other studies have found a variety of other rapidly evolving sperm–egg proteins in different species.
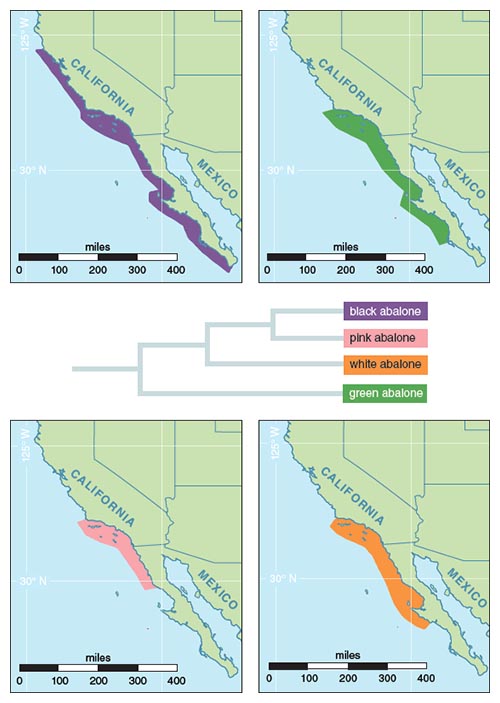
Illustration by Barbara Aulicino.
In particular, researchers in the Vacquier and Swanson labs are using the abalone system to study species specificity of sperm and egg binding. Historically, abalone species lived in overlapping boundaries across the coast of the western United States. They provide an ideal system for studying reproduction and species specificity, because even though species of abalone have overlapping ranges and spawning times, there is very little hybridization between species. This fact indicates that there must be some type of species specificity happening at the sperm–egg level. Researchers can use a conservative test for rapid evolution to study changes in sperm and egg genes (Figure 3). Indeed, we see that rapid changes have led to the extreme diversification of reproductive genes, even within closely related species of abalone (Figure 4). Why are reproductive genes so diverse, in some cases evolving much faster than immunity and defense genes that are under constant pressure because of microbial attacks?
Every sexually reproducing organism has its own unique strategy for attracting mates of the opposite sex. Mating strategies may involve alluring chemicals emitted by one sex, extravagant coloration, extreme size, sexy vocalizations and many other oddities. The male bowerbird works hard to build a colorful nest of rocks, flowers and sticks to entice females. And just like its namesake, the male peacock spider of Australia raises his elaborately colored flaps and dances to attract female onlookers. The female porcupine, however, prefers a shower of urine from male porcupine suitors before choosing a mate. These bizarre strategies have evolved over time in a complex balance that includes surviving to adulthood (time to mate!) and being attractive to potential mate(s) to pass on genes to the next generation.
A trait beneficial for male attractiveness may not necessarily be beneficial for male survival. For example, the male peacock has a colorful display of feathers irresistible to female peacocks, but which may attract undue attention from predators. The unwieldy tail might cause its demise. To boot, males and females may not have the same interests. Even if it is in the female’s interest to pick males that look like they have the best sperm, it is not necessarily in the males’ interest to look the part. Nevertheless, the process of sexual reproduction passes both sexes’ genes to their offspring indiscriminately. This could create a situation in which there is conflict between male and female reproductive genes. Sexual conflict happens when males and females are in an evolutionary arms race for optimal reproductive potential. Because two parties are involved, what is good for one may not be good for the other. For example, males may be evolving sperm that are better and faster at fusing with the egg than all the other sperm, whereas females may be counteracting these more efficient sperm by evolving eggs that resist quick fertilization to prevent polyspermy.
From the standpoint of passing on the most and best genes, is it better to be a promiscuous Casanova or Cleopatra, or, as in Jane Austen’s Pride and Prejudice, a faithful Mr. Darcy or Elizabeth Bennet? In promiscuous species, females mate with many partners during their estrous, or fertile, period. In monogamous species, females mate with one partner during their estrous period—so serial monogamists are included. Depending on a variety of factors, such as availability of resources, promiscuity or monogamy could be adaptive for a species. In highly promiscuous systems, males are free to sow their wild oats with many different females, giving them high reproductive potential. Although females don’t have as high of a reproductive potential as males, because they are limited by high female investment in eggs and young, they can maximize the continued proliferation of their genes by choosing the “perfect” mate(s) to fertilize their egg(s).
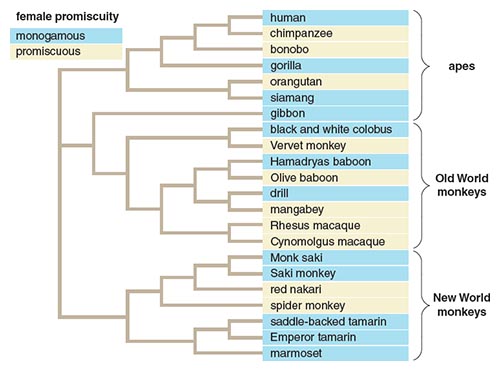
Illustration by Barbara Aulicino.
Because of their diverse mating systems, primates provide an excellent system to study how mating system differences have affected reproductive trait and protein evolution (See Figure 5). Even between closely related species, such as humans and bonobos, mating systems differ dramatically. Humans are mostly monogamous, and bonobos are some of the most promiscuous primates—a female bonobo may mate up to 50 times with different males during a single estrous period. Many studies have shown that in more promiscuous mating systems, size differences between the sexes, testes size in males, complex genital morphology and other morphological, behavioral and molecular traits areexaggerated. Richard Prum and colleagues at Yale University have shown that the complex maze of the female Pekin duck’s vagina may have evolved so that she could confound sperm duds and select only the best of the best sperm from males she has mated with. The corkscrew-shaped male Pekin duck’s penis enables him to penetrate the female, navigate the vaginal maze, and deposit his sperm in an advantageous location (See Figure 6). Work in Prum’s lab demonstrated that nonoptimal spirals in a duck’s penis have been shown to be less compatible with a female duck’s vagina. Thus, promiscuity and competition between males influence the evolution of complex sexual structures.

Image from P. L. Brennan, et al. Proceedings of the Royal Society B 277:1309.
Why is there so much diversity in reproductive traits and genes? Sexual selection favors genes that give a reproductive advantage, and these genes can increase over time in a given population. Sexual selection should not be confused with Charles Darwin’s famous theory of natural selection, which emphasizes the survival of the fittest. In our peacock example, natural selection would not favor the male peacock tail, because it decreases survival by increasing predation, but sexual selection through female preference favors an otherwise unfavorable trait. Although Darwin briefly discussed sexual selection in On the Origin of Species, modern interpretations of sexual selection group it into two parts: precopulatory sexual selection (before sex or mating occurs) and postcopulatory sexual selection (after sex occurs). Precopulatory sexual selection can occur within a sex (or between males) or between sexes (between males and females, see Figure 7). An example of within-sex competition is when males combat over access to females, as occurs in gorillas and big-horned sheep. The competition between males can increase the occurrence of “weaponry” in those males, such as antlers, horns, strength and body size. Precopulatory sexual selection between the sexes can occur when the female chooses her mate based on certain male qualities that are either behavioral or decorative. Behavior might include mating songs or dances, and decorative features include coloration or shape changes. For example, the male sage grouse has sacs on his chest that he inflates during the mating season to attract females. Change in such morphological features is driven by molecular changes within genes.
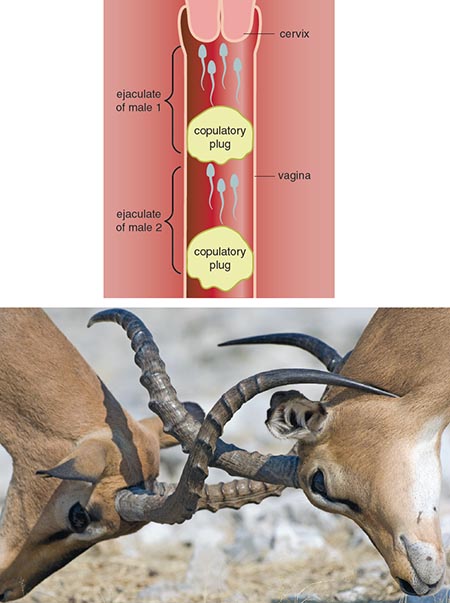
Illustration at top by Barbara Aulicino. Photo at bottom, Tony Camacho/Science Photo Library.
Why are we interested in these differences in mating strategies and reproductive traits and genes? The short answer is that many of these changes may have led to the evolution of new species in the past or could lead to the evolution of new species in the future. Studying these changes will lead to better understanding of the oddities seen in nature, such as outrageously long sperm and spiral-shaped penises. Many of the rapidly evolving reproductive genes are involved in more direct reproductive functions, such as sperm–egg binding, semen coagulation and fertilization. Although we do not know all the molecular mechanisms for sperm–egg interactions in every species, the puzzle pieces of how a sperm finds and fertilizes an egg are beginning to come together.
Everyone learns in basic biology that the sperm has to reach the egg for fertilization to occur. It sounds simple enough. Among internal fertilizers, the male(s) will deposit semen into the female’s reproductive tract, and the race to fertilize the egg begins. What about in external fertilizers where the sperm and eggs are released into the open environment? In both external and internal fertilizers, the egg releases chemicals to which sperm are attracted, and the millions or billions of sperm ejaculated or broadcasted follow this candy trail toward the egg. In a normal human male, it’s quite surprising how many sperm are either dead or abnormal on ejaculation. Many evolutionary biologists have proposed that some sperm may have alternative functions during fertilization: Kamikaze sperm were thought to reduce the chances of rival sperm fertilizing the egg by blocking and killing them with enzymes. This notion has not panned out in subsequent studies in humans but is still an interesting hypothesis that might be relevant in other species. Simone Immler’s lab at Uppsala University in Sweden showed that the hooked shape (Figure 1, middle of top row) of rodent sperm increased sperm swimming speed because the sperm hooked together to form clumps, which swam faster in a group than lone sperm. So maybe sperm work together for the good of the group.
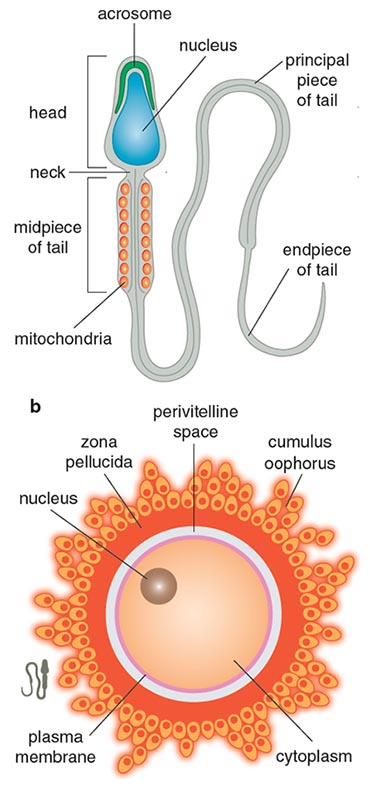
Illustration by Barbara Aulicino.
The basic structures of sperm and eggs are similar across mammals and invertebrates (See Figure 8). Each sperm has a compartment called the acrosome that contains digestive enzymes. In mammals, sperm fully mature in the female reproductive tract, where certain molecules trigger them into full motility. Sperm are not alone on their journey; fructose-rich seminal fluid accompanies the sperm and provides a protective and nutritious environment in the female reproductive tract. Seminal fluid neutralizes the acidic vaginal environment to make it habitable for sperm. Seminal fluid proteins also have antimicrobial functions that may be important for avoiding pathogens. In fruit flies, proteins in seminal fluid change female behavior after mating. In experiments conducted by Mariana Wolfner and her collaborators at Cornell University, specific seminal fluid proteins were “knocked down,” or rendered inactive, in a group of females during mating. The females with unaltered semen deposited in their reproductive tracts lost interest in remating with other males and tended to lay more eggs than the females with “knocked-down” semen. This difference occurs because there are proteins in semen that facilitate egg-laying and postmating behavior in females, although the exact mechanisms remain unclear.
As Emily Martin of New York University has made clear in her study of language used to describe fertilization, many textbooks and articles describe the egg as passively awaiting the arrival of the sperm. But the egg has its own journey: Once mature, it will leave the ovary and travel into the fallopian tubes. The egg surrounds itself with multiple barriers (shown in Figure 9), including a collection of follicular cells called the cumulus oophorus; the glycogen-rich matrix of the egg envelope (or zona pellucida in mammals); and a plasma membrane enclosing the cytoplasm and nucleus of the egg. These barriers protect the contents of the egg but also slow down any sperm trying to penetrate the egg. When more than two sperm fuse with an egg, it results in cell death. Polyspermy, the fusion of multiple sperm with an egg, causes an unbalanced number of chromosomes, which eventually leads to disintegration of the cellular bodies. External fertilizers may have additional outer barriers to provide further protection from the environment.
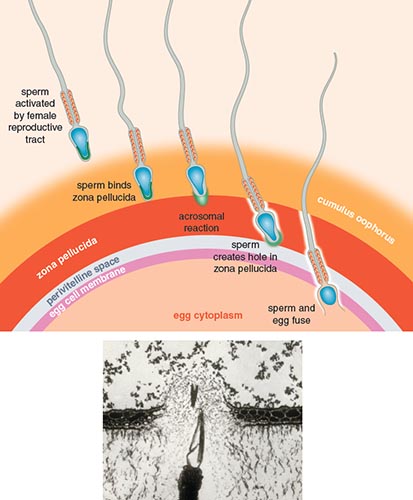
Illustration by Barbara Aulicino. Image at bottom from N. Kresge, et al., Bioessays 23:95.)
The sperm and egg meet. Then what happens? The first and most well-studied step in the fertilization process is the binding and passage of the sperm through the egg envelope. Many molecules on the sperm head and the egg’s outer surface facilitate binding, but few of these molecules have been identified. Abalone are used in many reproductive studies because as external fertilizers, their gametes are abundant and easily collected. In abalone, two interacting proteins have been identified on the sperm and egg, lysin and vitelline envelope receptor for lysin (VERL), respectively. The sperm protein lysin is found in the acrosome. Lysin is released when the sperm acrosomal contents are dispensed after binding to the egg in a process called the acrosome reaction. Lysin is then able to bind to VERL to create a hole through the egg envelope so that the sperm can pass through.
In other organisms, a variety of candidate proteins involved in binding the egg envelope have been proposed, but sperm proteins interacting with egg proteins only have been verified in abalone and sea urchins. Surprisingly, even in humans, no conclusive interacting sperm–egg proteins have been identified, despite the zona pellucida being well characterized. The proteins ZP3 and ZP2 are proposed to play an important role in binding sperm and are modified by parts of sugar molecules, which may be an important strategy for the egg to block nonoptimal sperm from binding. The sperm protein ZP3R/sp56 is proposed to bind with ZP3, but evidence for this binding remains contradictory. Protein complexes and parts of sugar molecules on the egg envelope may be involved in sperm–egg binding.
In the next step to fertilization, the sperm fuses with the egg plasma membrane. This step does not seem as chemically specific as the sperm binding to the egg envelope. For example, mouse sperm are able to bind and fuse with human eggs when the zona pellucida is removed. (Of course, the fused mouse sperm and human egg are nonviable.) In humans, the egg protein CD9 is proposed to fuse with sperm protein Izumo. The interaction mechanisms between these proteins remain elusive, but both CD9 and Izumo show evidence of rapid evolution. Genes involved in sperm–egg fusion may be quite diverse between taxa. Once the sperm and egg have fused, the resulting cell is called a zygote. The zygote undergoes rounds of mitosis and cellular specialization until an offspring is born.
Studying rapid evolution brings into focus candidate genes that are changing quickly—change that may be important to successful reproduction or the rise of new species. Not all organisms can be brought into the lab and studied intensively. Detecting rapidly evolving sperm and egg proteins with DNA sequencing enables the study of organisms that cannot easily be included in experiments, such as humans and other long-lived organisms. As mentioned earlier, sexual selection influences egg and sperm diversity, as well as rapid evolution in reproductive proteins. In any given case of rapid evolution in a gene, natural selection and sexual selection may be acting alone or in concert with each other. Some hypothesized reasons for rapid evolution in reproductive genes include scenarios called sperm competition and sexual conflict.
Sperm are competitive. With millions of sperm trying to get to the egg first, there is no doubt that subtle changes in shape or energy stores will change a sperm’s likelihood of successful fertilization. Adaptations to increase a male’s chances of reproductive success are particularly important within promiscuous mating systems. Sometimes more is better. In some promiscuous species, males with more sperm and greater testes size have better chances at fertilizing multiple females. Males have evolved various tactics to reduce the chances of another male’s sperm from fertilizing the egg, such as mate-guarding and copulatory plugs. One of the most promiscuous primate species is the bonobo. The bonobo has large testes size relative to body weight, and a thick copulatory plug will form from seminal fluids in the female’s reproductive tract after mating. By contrast, the king-of-the-jungle gorilla has tiny testes and a low sperm count, because the male gorilla has virtually no sperm competitors. After physically defending his territory and harem of females, the male gorilla will mate with all the females, under no pressure from other males.
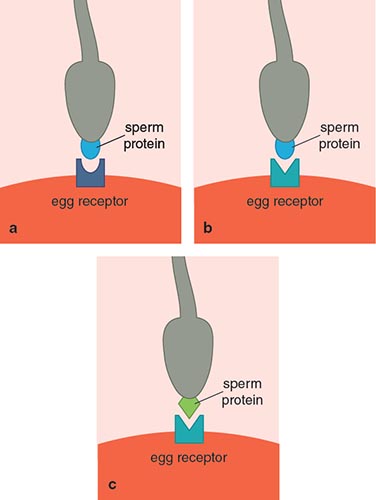
Illustration by Barbara Aulicino.
As mentioned earlier, a more competitive sperm is not necessarily to the female’s advantage. She may be looking for other qualities in a mate, or she may want to prevent polyspermy. Dissimilarities between the goals of the egg and those of the sperm may lead to sexual conflict. For example, the egg may be better off if the number of sperm that can enter is slowed through a mutation in an egg envelope protein. In such a situation, only sperm with a compatibly shaped protein would be able to bind and bypass the egg envelope. In turn, the sperm adapt to be more compatible, and thus competitive, with this new protein. We can think of this example as a lock and key, where the egg envelope is the lock and the sperm’s interacting protein is the key (See Figure 10). By changing the shape of the lock, the egg eliminates the number of keys that fit (or sperm that bind), because all it requires is one sperm, and no more than that. In response to the lock change, sperm evolve to change the shape of the key to counteract the blocking strategy of the egg. All of these changes leave a mark on reproductive genes. Such interacting lock and key proteins should be changing rapidly. Research has shown that lysin and VERL are rapidly evolving in abalone populations, and evolutionary patterns that indicate sexual conflict may be occurring.
In addition to the hypotheses of sperm competition and sexual conflict, there are a variety of other hypotheses to explain the evolution of sexual traits and genes. In plants, the ability to recognize self from nonself plays an important role in fertilization, because self-fertilization will result in less diverse offspring than fertilization with pollen from another individual. Many genes with reproductive roles also have antibacterial and immune functions, which indicate that the threat of microbial attack on the sperm or egg may be a major influence on rapid evolution during reproduction. Scenarios also exist where changes in a gene do not have a big effect, or where errors during DNA replication result in duplicating a gene on a chromosome, and as the twin genes change over generations, they specialize in their function. All or some of these pressures can act on a species’s reproductive characteristics, and researchers use molecular evidence to disentangle the various hypotheses.
Advances in genomic and proteomic technologies have provided new insights in reproductive research. Genomic technology enables us to sequence the entire genetic blueprint of organisms, from bacteria and viruses to platypuses and humans. The genome is the information storehouse for our bodies and encodes all the information we need to function. If DNA is the storehouse, then proteins are the workers that make things function. Proteomic technology such as mass spectrometry provides tools to identify proteins in specific tissues or cells and confirms the types of interactions that occur. The increasing ease of genomic sequencing has made many genomes available to test evolutionary hypotheses and study interesting organisms. Proteomic methods have enabled researchers to identify new proteins in sperm, egg, seminal fluid and follicular fluid, which may be important for identifying the causes of infertility. Our lab is using both proteomic and genomic technology to study evolution in reproductive proteins in abalone, plants and primates. My research in primate reproduction will elucidate how promiscuity and sperm competition are affecting the evolution of seminal fluid proteins. Further work using these methods will enable the identification of interacting sperm–egg proteins and the functional and structural characterization of reproductive proteins. We are only at the tip of the iceberg in understanding how sex influences biological diversity.
Click "American Scientist" to access home page
American Scientist Comments and Discussion
To discuss our articles or comment on them, please share them and tag American Scientist on social media platforms. Here are links to our profiles on Twitter, Facebook, and LinkedIn.
If we re-share your post, we will moderate comments/discussion following our comments policy.