Ancient Wollemi Pines Resurgent
By Stephen McLoughlin, Vivi Vajda
Ten years after its discovery, a vanishingly rare tree from the Cretaceous Period is a scientific darling and may soon become a commercial success too
Ten years after its discovery, a vanishingly rare tree from the Cretaceous Period is a scientific darling and may soon become a commercial success too

DOI: 10.1511/2005.56.540
In September of 1994—springtime in Australia—three adventurers spent a Saturday hiking, rock climbing and rappelling through the rugged sandstone ravines of Wollemi National Park, a wilderness area beyond the northwestern outskirts of Sydney.
After abseiling down into one narrow canyon, David Noble, the leader of the group and an officer with the National Parks and Wildlife Service of New South Wales, noticed a patch of large, peculiar trees. A veteran of hundreds of expeditions in the park, Noble puzzled at the distinctive yet unfamiliar trees before him. Almost as an afterthought, he collected a few leafy twigs and stashed them in his pack.

Shane Pederson
Some days later, Noble showed the twigs to Wyn Jones, a naturalist with the National Parks and Wildlife Service. Jones looked at the dried-out leaves and suggested that they belonged to a fern or cycad—until Noble told him that they came from a tree that was 35 meters tall! The men consulted with another expert, botanist Ken Hill from the Royal Botanic Gardens in Sydney, but he also failed to recognize the specimen.
To solve this mystery, Noble, Hill and another biologist, Jan Allan from the nearby Mount Tomah Botanic Gardens, set out the next month to photograph the plants and collect leaves, bark and fertile material that would enable a precise identification. Only then did the investigators realize that they had stumbled upon not only an unknown species, but also a tree outside any existing genus of the ancient Araucariaceae family of conifers. The strange trees before them may have been some of the rarest plants on Earth. The find was all the more astonishing given the towering size and the location of the trees—less than 200 kilometers from Sydney, a city of more than four million people.
Within months, media outlets around the world reported the discovery of a "tree from the age of the dinosaurs," bringing to mind Arthur Conan Doyle's Lost World. The mystique was further enhanced by the immediate decree of tough restrictions on public access to the only known specimens, about two dozen individuals in a tiny grove. Such news captured the imagination of the scientific community and general public alike. Indeed, the buzz surrounding these exceptionally rare plants has been a crucial component of their survival.
The high profile of the wollemi pine among paleobotanists and ecologists has resulted in more than 35 scientific papers in the past 10 years. The species has also been featured in popular books, newspaper articles, television documentaries, posters, postcards and even songs. Tens of thousands of Web pages mention wollemi pines, a number that grows rapidly each week. Most importantly for the survival of the trees, conservationists have worked with commercial interests in an effort to market the plant worldwide as a horticultural stalwart; the first shipments are set for late 2005 or early 2006.
The wollemi pine was described and given its official name, Wollemia nobilis, in a short article published in a 1995 volume of Telopea, the botanical journal of the National Herbarium of New South Wales. The species epithet is indicative of the noble stature of the tree and the name of its discoverer, David Noble. The genus name is derived from its place of discovery, obviously, but wollemi—an Australian aboriginal word meaning "watch out, look around you"—is also an apt warning when traversing the tangled maze of sandstone escarpments surrounding the plant's habitat.

J. Plaza, Botanic Gardens Trust
Describing the tree as a "pine" is a slight misnomer, as it is not closely related to the true pines, which properly belong to the genus Pinus and are typically found in the Northern Hemisphere. Apart from Wollemia, only two other living genera are included in the family Araucariaceae: Araucaria and Agathis. The former includes trees that are important to the timber industry in Australia, such as the bunya bunya and hoop pines, as well as statuesque ornamental trees such as the Norfolk Island pine and the monkey-puzzle tree, a native of the Andes. Modern Araucaria species grow in New Guinea, northeastern Australia, and southern South America, but the genus is most diverse in New Caledonia, a large island in the South Pacific east of Australia, on which at least 13 species can be found. The other family member, Agathis, is represented by the kauri pines, which range through northern New Zealand, northeastern Australia, the Southeast Asian archipelago, New Caledonia and Fiji. With five species, New Caledonia also constitutes the principal stronghold for Agathis.
Russell House
The relative abundance of Araucariaceae remains in the fossil record has allowed paleobotanists to understand much of the family's past distribution and evolution. Although Araucariaceae are found predominantly in the Southern Hemisphere today, this range appears to be relictual—that is, only a remnant of a once-more widespread population. The fossil record indicates that the family had a global distribution in the Jurassic (199-145 million years ago) and Cretaceous periods (145-65 million years ago). Indeed, some of the oldest such records date from more than 200 million years ago and occur as far away as Greenland and Sweden. A time-traveler visiting the coastal woodlands of Wyoming during the Middle Jurassic or the forests of southern England during the Eocene might well have rested beneath the branches of an Araucariaceae family member.

Photographs courtesy of the authors
In the past ten years, taxonomists have spent considerable effort researching the phylogeny of the wollemi pine, but the precise relationships within the Araucariaceae remain uncertain. It is not yet clear whether Wollemia is more closely related to Araucaria or to Agathis, or indeed if those two are more closely related to each other than either is to Wollemia. Studies of living species within these genera have suggested alternative relationships depending on which molecular markers are used in the analyses. Similarly, certain aspects of the wollemi pine's morphology (for example, its spiny cones and rough bark) are more similar to Araucaria, but other features (for example, its seeds that are shed independently of the cone scales) are more similar to Agathis. To resolve the order of these branches of the family tree, future studies will undoubtedly continue to use a more comprehensive combination of shape (morphological characteristics) and substance (genetic markers).
Much research has focused on the conservation status and ecology of the wollemi pine. Only three small copses are currently known, and these all occur within neighboring, shaded, permanently moist pockets of rainforest. The mature wollemi pines stand over a canopy of coachwood (Ceratopetalum), sassafras (Atherosperma), lily pily (Syzygium), and possumwood (Quintinia). Although large numbers of wollemi pine seedlings occur in the groves, it seems that few if any of these ever grow to maturity. Genetic studies of the adult plants show virtually no variation within the populations, suggesting that the individual plants are all natural clones. Unlike other members of its family, the wollemi pine shows a strong coppicing habit—younger stems emerge from the base of the tree and replace older trunks with time. As a consequence, tree-ring analyses of individual stems will not reveal the longevity of a plant as a whole. It is possible that some plants within the natural stands are many hundreds of years old, although any one trunk will be much younger.
Other investigators have focused on the biochemistry of the wollemi pine and associated organisms within its ecosystem. One such study revealed that a so-called endophytic fungus, which grows inside the plant itself, produces the chemical taxol—an important cancer-fighting agent. Such discoveries raise the hope that Wollemia will have other pharmaceutical or industrial uses. With scientists still ignorant about much of the fundamental biochemistry of the wollemi pine, its potential, like that of most other Australian plants, remains unrealized. Indeed, many of the microorganisms recently isolated from the soil of the wollemi pine's habitat have not even been formally named, let alone analyzed for active chemical compounds.
Since the trees' initial discovery, government authorities have given prime consideration to preserving the natural stands of these majestic trees, which faced two significant threats. Firstly, widespread reports of the significance and extreme rarity of the wollemi pine made it an instant target for unscrupulous collectors. Secondly, the plant's lack of genetic variability, coupled with the susceptibility of many native Australian plants to attack by foreign pathogens, made it highly vulnerable to diseases carried by visitors. These risks dictated a critical importance to the control of human traffic to the site. Fortunately, the plants were discovered within the bounds of a national park, and managers imposed a strict embargo on the whereabouts of the natural populations. In addition, the state government of New South Wales invoked a public exclusion zone around the trees and threatened a heavy fine and prison sentence for anyone who damaged them. Had the wollemi pines been discovered on private land, their future would have been less certain. Even their present security owes as much to good fortune as good planning: The region only became a national park in 1979. Its preservation as a wilderness area so close to Sydney was principally due to a rugged terrain that was unsuitable for development. The area is so impenetrable that it acted as a physical barrier to colonial expansion for a quarter-century after Sydney Cove was first settled by Europeans.
In the geological arena, the discovery of the wollemi pine has helped to solve several paleobotanical riddles, and the fossil record has, in turn, elucidated much about the ancient history of this plant.

Photograph courtesy of the authors
Just as the wollemi pine was discovered, one of us (McLoughlin), with colleagues Andrew Drinnan and Andrew Rozefelds of the University of Melbourne, submitted an article to the Memoirs of the Queensland Museum that described an assemblage of 100-million-year-old fossils from the broad, semi-arid plains of western Queensland (some 1,500 kilometers northwest of Sydney).

Photographs courtesy of the authors
This assemblage contained two main types of twigs from large cone-bearing trees: One group of slender shoots had long, narrow leaves that were twisted at the base to form a flattened arrangement like a palm frond. Another group of stout shoots had broad, short leaves arranged in four ranks that spiraled around the stem. Although tempted to describe these fossils as two distinct species, we noted that a few specimens had intermediate features, which suggested that these dissimilar shoots belonged to the same plant.
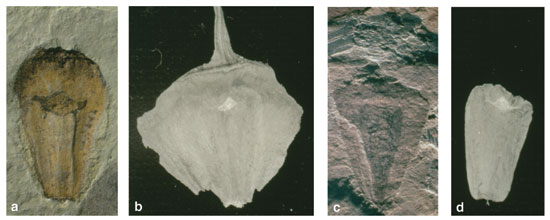
Photographs courtesy of the authors
When material from the wollemi pine came to hand a year later, we realized that the living plant closely matched the Queensland fossils. Those with thin branches and flattened leaves corresponded to juvenile wollemi shoots and the robust fossil branches with spiral leaves matched wollemi adults. Fossil pollen and seed cones from the region also looked like those of the modern wollemi pine. Since then, other paleontologists have discovered many similar fossils in western Queensland during recent excavations of dinosaur skeletons. Furthermore, the rocks of coastal Victoria (about 700 kilometers south of Sydney) also contain fossil leaves and cone scales similar to those of the wollemi pine, and these are even older than the Queensland specimens, dating to 120 million years ago—a time when Africa and South America lay together like spoons and Australia was still connected to Antarctica (the globe was a bit warmer then). These records show that wollemi pines were common for tens of millions of years on the Australian continent, growing in tall, moist forests alongside an abundance of flowering plants, ginkgos, cycads and ferns.

Photographs b and d courtesy of Wollemi Australia. Photographs a and c courtesy of the authors.
The pollen of the wollemi pine solved another fossil riddle. Palynologists—scientists who study pollen and spores—have known about a type of fossil pollen called Dilwynites (this is the name of its genus) in Australia and New Zealand since 1965. It was generally similar to the pollen of Araucaria but had a coarser, more granular coating. Paleobotanists assumed that the parent of this ancient pollen was extinct, as no living plant produced it. That assumption was disproved with the discovery of the wollemi pine, whose pollen perfectly matched the fossil specimens—here at last was the origin of the mysterious Dilwynites. By drawing inferences from the habitat of the wollemi pine, scientists have revised their estimates of the environmental conditions of ancient Australasia based on the distribution of Dilwynites.
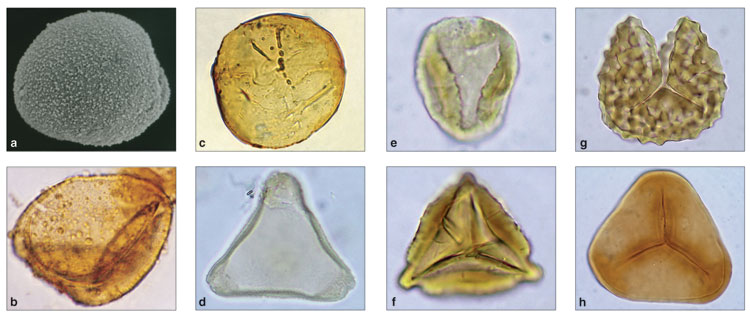
Photographs courtesy of the authors
Pollen grains disperse widely and make for durable fossils because of a waxy, enzyme-resistant coating made from a unique substance called sporopollenin. Consequently, the distribution of ancient, fossilized pollen provides the best insights into the ancient history of Wollemia. The earliest fossilized leaves and cones that resemble those of the wollemi pine are from eastern Australia, contained in rock from the Early Cretaceous period; Dilwynites is clearly present in the region by 90 million years ago, and it appears in New Zealand by 70 million years ago. At this time, the Tasman Sea between Australia and New Zealand was much narrower, which probably permitted a greater interchange of species (including the wollemi pine) than at present.
Based on high-resolution records of fossil pollen from New Zealand, Wollemia and most other forest plants experienced a short-term dieback after the asteroid impact that drove the dinosaurs extinct and ended the Cretaceous. This collision, which occurred on Mexico's Yucatan Peninsula 65 million years ago, ejected huge quantities of rock that fell back to Earth as flaming meteorites, igniting a global firestorm and leading to an "impact winter" as soot and dust in the atmosphere blocked out the sun. However, Wollemia apparently survived the cataclysm in small refugia or as seeds protected within the soil and soon recovered its place in the moist, temperate forests of Australasia. Pollen in sediments from Antarctica's continental shelf indicates that the species reached that continent more than 50 million years ago. Wollemia pollen peaked in abundance during the Paleocene-Eocene epochs (65-34 million years ago), then declined steadily throughout its range. This decrease corresponded to dramatic global cooling and Australia's continental drift northward into drier latitudes. The most recent known Dilwynites was recorded in two-million-year-old sediments from beneath the seafloor of Bass Strait (between Tasmania and mainland Australia)—until the discovery of the modern wollemi pine!
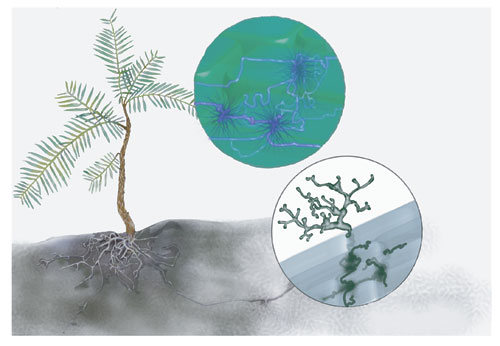
Russell House
The special characteristics of the tree's habitat appear to have facilitated its survival in such small groves. Hidden in narrow sandstone ravines, the wollemi pine enjoys consistent humidity and moist soils, which suit both the plant and the mycorrhizal fungi that live in association with its roots. Like almost all Australian plants, Wollemia depends heavily on such symbiotic fungi to penetrate hard ground and take up nutrients from the continent's notoriously infertile soils. However, those that coexist with the wollemi pine are unlikely to thrive in the thin, drier soils of the surrounding plateaus.
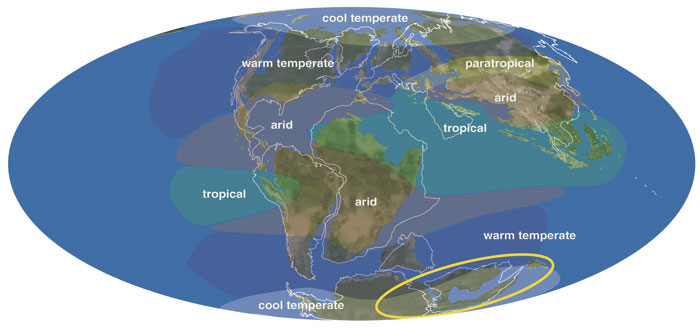
Adapted from C. Scotese's PALEOMAP Project (http://www.scotese.com). Illustration by Russell House.
The location and pattern of growth of Wollemia also provide some protection from fire. The tree's coppicing habit probably helps it survive the occasional, small fire, although it wouldn't do much good against the intense firestorms that regularly ravage Eucalyptus-dominated forests on the tops of the sandstone plateaus. But within its moist ravines, the wollemi pine is largely protected from such fires.
The numbers and types of fossil spores and pollen from across Australia indicate that the continent became progressively drier through the latter half of the Cenozoic, beginning about 30 million years ago. This trend was particularly pronounced during the Pleistocene, beginning 1.8 million years ago, when continents in the northern hemisphere experienced cycles of widespread glaciation. In Australia, microfossil assemblages from the Pleistocene also contain many more particles of charcoal. Hence, these two factors—a drying landscape and more frequent fires—appear to have been the main reasons for the gradual decline of the wollemi pine through the Cenozoic Era.
Although it took a few weeks for authorities to realize that they had discovered a large, new species of conifer, the wollemi pine soon became a celebrity. Potted specimens were taken on a national tour of Australian botanic gardens, and several were planted in those gardens, protected from over-enthusiastic plant collectors by steel security cages. Within three years, the plant even took up residence in Kew Gardens in London. Unfortunately, the scant number of seedlings made wider distribution impossible, so the prospects for the wollemi pine remained uncertain.
Fortunately, the Botanic Gardens Trust of Sydney quickly recognized the tree's horticultural potential. They licensed Wollemi Australia, a joint venture between the Department of Primary Industries (Forestry) in Queensland and Birkdale Nursery in Brisbane, to propagate the plant on a commercial scale and market it to an international audience. Conservationists believed that this effort was an important strategy for the long-term survival of the species. Not surprisingly, much early research focused on understanding the germination of wollemi pine seeds. This work revealed that the tree sheds most of its seeds in late summer and early autumn, and that they germinate fastest in relatively warm conditions (24-30 degrees) when exposed to sunlight. However, propagation by seed stocks would be slow, which risked the loss of public interest, so horticulturalists turned to tissue culture as a means of growing thousands of young plants for distribution. The organization aims to begin selling the plant near the end of 2005 or in early 2006.

J. Plaza, Botanic Gardens Trust
If this international marketing plan succeeds, the wollemi pine may transform from one of the rarest plants on Earth to one of the most widespread ornamentals in temperate to subtropical regions. However, it won't suit everyone's garden: Many back yards won't accommodate a tree that reaches 35 meters at maturity—especially one that tends to shed whole branches rather than individual leaves. It's not an easy plant to grow either, as juveniles are extremely susceptible to attack by pathogenic microorganisms such as Botryosphaeria species and Phytophthora cinnamomi, the cause of root rot. More positively, the wollemi pine is likely to be suitable as a patio or indoor plant given its tolerance for shaded conditions.
If the wollemi pine does spread rapidly across the planet, it may benefit science in one last, unexpected way. Australian palynologists and archeologists commonly use the first appearance of Pinus pollen in a succession of sedimentary layers to synchronize the recent geological and chronological records. European settlers, who first arrived in Australia in 1788, changed the landscape so rapidly and introduced so many of their homeland plants that the appearance of pollen from non-native true pines marks a definitive horizon for colonization. In similar fashion, Wollemia's imminent commercial invasion of the rest of the world may make its distinctive, granular pollen a valuable marker of the dawn of the 21st century for archeologists of the future.
Although the wollemi pine has yielded scientific insights in several fields, its most significant effect has been to raise the profile of the natural sciences amongst the public. It was the first new species of conifer to be found since the dawn redwood (Metasequoia glyptostroboides) was discovered in China in 1948. Its existence has emphasized that humans still have much to learn about our natural environment and that a great number of species—including some very large ones—have yet to be discovered. This point was highlighted again by the discovery in 1999 of another conifer, the golden cypress (Xanthocyparis vietnamensis), in Vietnam. The attention paid by mass media to these discoveries has shined a welcome light on the desperate straits of rare and endangered species around the world. In this case, at least, the danger of extinction has largely passed: The Wollemi Pine Conservation Club has been chartered to safeguard the future of the species, and royalties from sales of the plants will support conservation of the wollemi pine and other threatened species for years to come.
So perhaps in the future, as the wollemi pine penetrates the international market, Santa Claus may find himself stacking presents beneath a new type of Christmas tree—one that escaped extinction by the barest of margins and, with a little help, broke out of its rainforest refuge to re-colonize the world.
Click "American Scientist" to access home page
American Scientist Comments and Discussion
To discuss our articles or comment on them, please share them and tag American Scientist on social media platforms. Here are links to our profiles on Twitter, Facebook, and LinkedIn.
If we re-share your post, we will moderate comments/discussion following our comments policy.