Pathogens, Host-Cell Invasion and Disease
By Erich Gulbins, Florian Lang
Invading pathogens can co-opt even the cells of the immune system. New anti-infective drugs may arise from an understanding of this chemical warfare
Invading pathogens can co-opt even the cells of the immune system. New anti-infective drugs may arise from an understanding of this chemical warfare

DOI: 10.1511/2001.34.406
Since the dawn of civilization, infectious diseases have shaped human history. In the Middle Ages, Europe lost up to a third of its population to plague outbreaks. The Spanish flu outbreak during the winter of 1918–19 killed more people—between 25 and 40 million—than did World War I.
It was not until 1941, with the development of penicillin, that science could offer a potent weapon against infection, at least against bacterial diseases. But modern medicine is far from having won the war against pathogens, organisms that cause disease. Infectious diseases, including AIDS and malaria, are a severe health problem in the Third World. And in the developed world, bacteria are becoming increasingly resistant to the antibiotic drugs available to combat them.
Tony Brain/Science Photo Library
To develop new treatments against infectious diseases, whether caused by viruses, bacteria or higher microorganisms, it is important to better understand the nature of infection—the mechanisms and strategies that pathogens use to invade the body's cells as well as overcome the body's defenses against them. New approaches are needed that do not trigger evolutionary countermoves by the pathogens. Such strategies, we hope, might arise from a better understanding of the ways pathogens invade host cells and evade the immune system—an area of research neglected while antibiotics provided easy answers, but clearly essential to providing new weapons for the continuing battle against infectious disease. Recent research has therefore been concentrating on the mechanisms that infectious agents use to gain entrance to their target cells as well as the strategies that the host evolves to prevent them from doing so. Using this knowledge, drug developers will eventually be able to develop new anti-infective medications that prevent pathogens from penetrating host cells. Investigators hope that strengthening the host's defenses, rather than attacking the pathogens directly, will provide therapies against infectious disease that are more durable over the long term than those offered by antibiotics.
Organisms have always been attacked by pathogens; the species that have survived, including us, are those that have evolved means to fend off their attacks. Likewise, pathogens have developed means to overcome these protective measures, forcing the evolution of even more powerful host defenses. Over millions of years, the arms race between pathogenic organisms and animals eventually created the mammalian immune system, which includes a wide variety of highly specialized compounds (toxic proteins that attack foreign cells marked with antibodies) as well as immune cells—the large macrophages of the blood and lymph, the natural killer cells, T lymphocytes and antibody-producing B lymphocytes. Immune cells patrol the bloodstream, various organ tissues, the surfaces of the intestines and the respiratory and urinary tracts.
No matter where a pathogen comes from—via the respiratory or the digestive tract, an injury or an insect bite—it faces an army of immune cells that are specialized to devour invading pathogens, shower them with toxic chemicals, punch holes into their cell membranes or bombard them with antibodies to mark them for destruction. In fact, this defense system is so effective that most people spend most of their lives free of infectious disease, despite daily exposure to countless viruses, bacteria and infectious eukaryotic parasites (microorganisms with nucleated cells, such as Plasmodium, the protozoan that causes malaria).
But pathogens, having the advantage of larger numbers, can overwhelm the host's immune system and often adapt more nimbly. Mutation and selection have provided them with mechanisms for circumventing or countering the immune system's attacks. Many pathogens, notably viruses but also many bacteria and eukaryotic microorganisms, invade host cells as a first step so that they are hidden from the immune system as they multiply. However, invasion is not a one-sided process; it often requires the active participation and cooperation of the host. The properties of both host and pathogen determine whether the pathogen can successfully establish an infection or whether it is killed by the host.
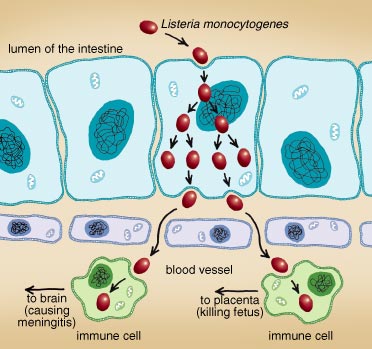
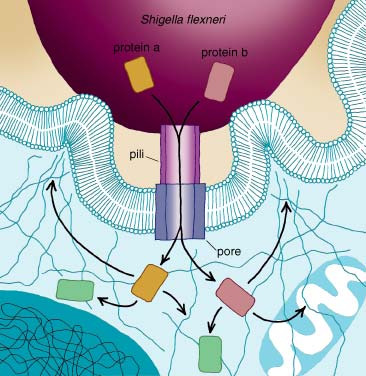
The immune system can detect alien organisms only outside cells or on the cell surface. This leaves the system vulnerable to attack from within its own cells. For instance, Shigella flexneri, a bacterium that infects people through feces-contaminated food and causes severe diarrhea and vomiting, first attaches to and then penetrates macrophages—immune cells that are specialized in devouring invading pathogens—as they patrol the intestines. Shigella multiplies inside the host cell, where it is unchallenged by the immune system. Eventually, the infected macrophage dies and releases new bacteria that spread to and infect nearby epithelial cells and cause destruction of the intestinal tissue.Listeria monocytogenes, a bacterium that causes meningitis and spontaneous abortion in pregnant women, first infects epithelial cells in the intestinal tract as it is taken up with contaminated food. The bacteria multiply and cross the epithelium to the bloodstream. It is in the blood that they penetrate the patient's immune cells, which then carry the infection all over the body. If those infected cells get into the brain and release the Listeria, they can cause meningitis, a life-threatening infection of the brain's surrounding membrane. If the infected cells travel through the placenta of a pregnant woman and release the pathogen in the fetus, the subsequent infection can kill the unborn child.
Tom Dunne
Another natural target is the lining of cells on the surface of the intestines or the lungs, another of the body's natural barriers against infectious microorganisms. All surfaces that are exposed to the outside—the skin, the intestines, the respiratory and urinary tracts—are lined with epithelial cells that are so tightly interconnected that no virus or bacterium can squeeze through. In addition, these surfaces are covered with immune cells ready to attack any invading alien structure. Many pathogens therefore infect these cells first to establish a bridgehead from which they can release their progeny into the tissues and blood vessels that lie on the other, inner side of the epithelial barrier. The bloodstream may then become the medium through which they can spread throughout the body and infect other tissues and organs.
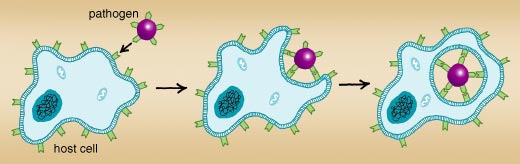
Pathogens have evolved a wide variety of mechanisms and strategies that enable them to enter body cells. However, a pathogen's invasion of a target cell often requires the active participation of the host. Indeed, the internalization of a pathogen involves the concerted interplay of many bacterial and host-cell proteins, and also involves the plasma membrane, the outer membrane that surrounds the host cell. The challenge for a pathogen, similar to the problem faced by a burglar cracking a safe, is to find a chemical code that starts the processes that eventually lead to its internalization by the host cell.
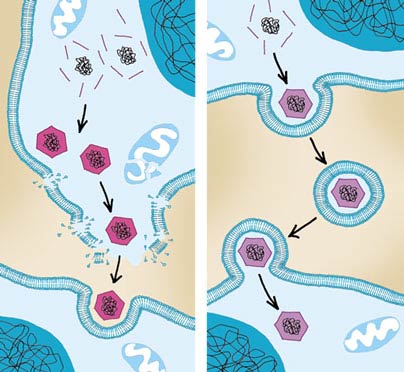
Internalization begins when the invading germ attaches to the host cell's plasma membrane and triggers the host cell to wrap its membrane around the invader. Eventually, when the pathogen is completely wrapped up by the cell membrane, this structure buds off to form a membrane-coated vesicle with the virus, bacterium or other microorganism inside. Listeria attaches to its target by displaying a so-called InlA (internalin) protein on its surface. InlA is specialized in making contact with a protein called E-cadherin on the surface of epithelial cells in the intestines. This initial contact triggers the movement of other host-cell surface proteins to the binding site; these reinforce the binding of the bacterium to the epithelial cell and help to stimulate the cytoskeleton. Another protein on Listeria's surface, InlB, contacts two surface receptors on the host cell, which in turn activate an enzyme called PI3-kinase in the cell. PI3-kinase starts a series of events in the cell, which eventually induce the changes in the cytoskeleton that are necessary to internalize the attached bacterium. Indeed, this enzyme plays a key role in the Listeria infection process; experiments with an inhibitor of PI3-kinase have shown that the bacterium is unable to enter the cell if the enzyme cannot be activated.
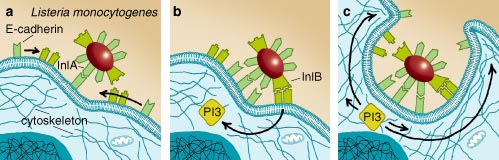
An important factor in this process is the cytoskeleton, a scaffold of various proteins in the cell that define and maintain the cell's form. It is the cytoskeleton that performs the initial ruffling of the plasma membrane and the subsequent changes needed to wrap the membrane around the pathogen. This is an intricate process, so an invading pathogen has to manipulate the host cell's biochemical machinery considerably to induce its internalization. This task is performed by specialized molecules on the bacterium's surface that attach to specific receptor molecules on the host cell's surface. These receptors are molecular switches that react to specific molecules in the outer medium and induce metabolic changes within the cell. By pressing the right buttons, sending the right signals to the target cell, an invading pathogen can thus start the necessary events in the host cell's interior.
Many viruses use similar mechanisms to get into their target cells. The human immunodeficiency virus (HIV) that causes AIDS, for instance, first attaches to several proteins on the host cell's surface, triggering its internalization. After the virus has been taken up by the cell, its hull dissolves and releases the virus's genetic material, which is used by the host cell's biochemical machinery to produce more virus particles.
Several other bacteria have developed an even more sophisticated mechanism for forcing entry into the host cell. They carry needle-like structures on their surface through which they inject into the host cell proteins that eventually trigger internalization of the bacterium. Shigella flexneri uses this mechanism, called a type III secretion system, to inject two proteins that interact with signal-transduction proteins and the host cell's cytoskeleton. The activation of those proteins finally results in the reorganization of the cytoskeleton and the formation of ruffles in the plasma membrane required to internalize the pathogen.
A variation of this process, called phagocytosis, is actually used as a defense mechanism against invading bacteria. Some immune cells, and also epithelial cells in the intestines and the respiratory tract, engulf invading pathogens, take them up and digest them by breaking them down into their constituents. In particular, macrophages are specialized in destroying pathogenic bacteria through this process. Yet some pathogens, such as Listeria or the plague-causing Yersinia pestis, are able to manipulate phagocytosis so that they survive.
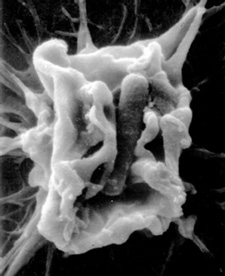
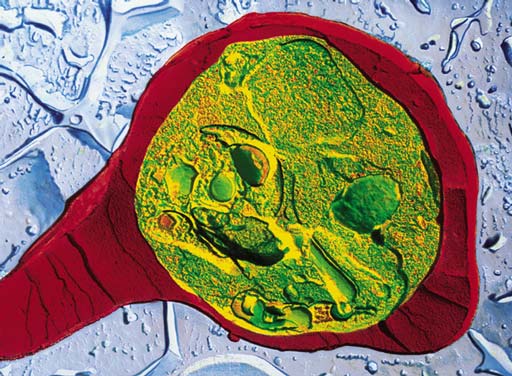
Electron micrograph courtesy of Pascale Cossart, Institut Pasteur; reprinted from Science 276:720, by permission of the American Association for the Advancement of Science.
For those pathogens, it is important to stop the machinery eventually leading to their digestion. When a macrophage engulfs a cell, the result is a newly created vesicle called a phagosome. The phagosome fuses with a lysosome, another vesicle that contains a high concentration of digestive enzymes specialized in breaking down biological molecules. Several pathogens, such as the tuberculosis-causing Mycobacterium tuberculosis, the leprosy-causing Mycobacterium leprae or Legionella pneumophila, the cause of Legionnaires' disease, have been shown to prevent or slow down the fusion of the phagosome with the lysosome. The ability to redirect the phagosome pathway is presumably important for their survival and replication in the infected cell.
The infection of a cell allows pathogens to take over the cell's biochemical machinery and produce offspring by using the nutrients that it finds in the host cell. Eventually, the production requires the delivery of additional nutrients at a rate that far exceeds the normal demand of a noninfected cell. Thus the pathogen has to manipulate the host cell's transport systems in the plasma membrane, not only to allow for this additional uptake of nutrients, but also to dispose of waste products that accumulate through the intense activity.
This manipulation is particularly obvious during the infection of red blood cells by the malaria-causing protozoan Plasmodium. Before infection, an erythrocyte is little more than a sack of hemoglobin proteins with a very low need for nutrients. Its membrane mainly contains transporter proteins that exchange bicarbonate ions, HCO3–, with the surrounding medium and maintain the cell volume through the exchange of other ions. Since the cell does not synthesize proteins, DNA or membranes, it has no need for amino acids, nucleic acids, lipids, vitamins and so forth. The replicating malaria pathogen, however, has an excessive requirement for all these nutrients. Furthermore, to fuel the replication process, Plasmodium needs large amounts of glucose; indeed, an infected erythrocyte takes up 40 to 100 times more glucose than a noninfected cell.
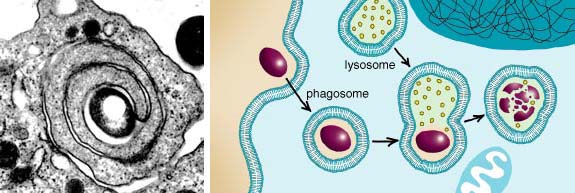
Micrograph courtesy of Pascale Cossart; reprinted from Cell by permission of Elsevier Science.
To gain access to the necessary nutrients, Plasmodium forces the host cell to alter the transport properties of its membrane by inducing the so-called new permeability pathway. The pathogen forces the infected cell to activate additional transport proteins in its plasma membrane that carry sugars, nucleic acids, membrane components and other substances from the intercellular medium into the cell. The necessity of the new permeability pathway for Plasmodium's survival and replication is illustrated by the fact that several inhibitors of this pathway eventually kill the pathogen while it still remains in the erythrocyte.
The essential nature of the new permeability pathway is not yet clear and is being intensely studied. Theoretically, the pathogen could express the respective transport systems itself and insert the proteins into the host-cell membrane. The advantage of this approach is that Plasmodium would not depend for its survival on the presence of adequate host-cell membrane proteins. The disadvantage, however, is that its proteins would be exposed at the membrane and thus would be recognized as alien structures by antibodies. Using the antibodies as markers, immune cells would sweep in and destroy the infected cell and the pathogen in it.
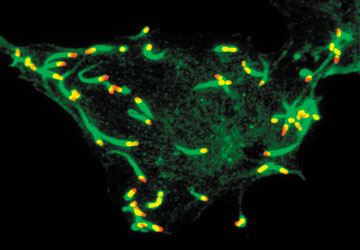
Immunofluorescence micrograph courtesy of Pascale Cossart.
Alternatively, Plasmodium could modify existing host-cell membrane proteins to turn them into the new permeability pathway. The advantage of this mechanism is that the cell would remain undetected by the immune system. Indeed, recent experiments indicate that at least part of the new permeability pathway results from modification of existing cell-membrane proteins by the pathogen. Because Plasmodium depends on creation of the new permeability pathway to proliferate in the infected cell, investigators are hoping to find new malaria drugs to block this pathway. There is little doubt that other pathogens that rapidly proliferate inside infected host cells have to modify the transport properties of these cells in order to gain access to nutrients. But not much is known so far about the changes in nutrient transport that these pathogens induce in their host cells.
As we mentioned above, penetration of the host cell depends not only on the properties of the pathogen but also on the properties of the host. Some individuals have assets that protect their cells from being infected by certain pathogens. Most important to an appropriate defense is the ability of the immune system to kill pathogens before they enter host cells. Consequently, older people with a weaker immune system, as well as patients with an impaired immune system—HIV patients or people with an organ transplant who have to take immunosuppressive medication to suppress rejection of the transplanted organ—are more vulnerable to infectious diseases. Also, the immune system's ability to "remember" pathogens and to mount a faster and stronger defense in case of reinfection confers a strong advantage over time. Young children are particularly vulnerable to many diseases because their immune system is encountering the invading pathogen for the first time.
In addition, some people may be more or less resistant to some pathogens because of their genetic makeup. For instance, a certain mutation in a gene that carries the information for a receptor protein on the surface of white blood cells renders people partly or fully resistant to HIV. HIV uses this protein to attach to and infect white blood cells; the mutation changes the properties of the receptor, so that HIV is less able to infect those cells. It is a rare mutation, but it will eventually become more common in Africa and South Asia because it gives a clear evolutionary advantage for individuals in those areas where AIDS is rampant.
Sickle cell disease is the result of a mutation that changes a single amino acid—swapping glutamate for valine—in hemoglobin, the oxygen-storing protein in red blood cells. A patient who receives this mutation from both parents is severely ill, because hemoglobin precipitates and deforms the erythrocytes as soon as it is deprived of oxygen. However, an individual who carries only one mutant allele does not suffer from the disease; rather, the mutation impedes the intracellular survival of Plasmodium when the person is exposed to the pathogen. It is not entirely clear how the mutated hemoglobin protects against malaria.
In any case, the malaria pathogen has apparently never been able to overcome this resistance through evolution. Thus, individuals carrying the mutation have better survival chances in those areas where malaria is endemic, particularly in Africa and Southeast Asia. As a result, the incidence of the sickle cell trait and of sickle cell disease is particularly high in those areas. Similarly, thalassemia and glucose-6 phosphate dehydrogenase deficiency confer some resistance to malaria. Again, those diseases are particularly frequent in malaria-infested areas.
However comfortable life in an infected host cell may be, invading pathogens must leave in order to spread to other cells or tissues. Many viruses and bacteria do this in a rather crude way, by destroying the plasma membrane. The host cell simply bursts, releasing the pathogens into the surrounding medium. This process eventually leads to partial destruction of the tissue, leaving in its wake the ruins of dead cells.
Other pathogens, among them many viruses, leave the cell by using a reversal of the initial integration process. New viruses produced by the host cell travel to the plasma membrane, where they are wrapped in a membrane vesicle that buds off into the surrounding tissue.
The sudden release of pathogens is particularly dramatic in the case of Plasmodium. All infected red blood cells rupture synchronously to release into the blood stream a large number of new pathogens, which infect other erythrocytes. This cycle is repeated every two to three days. Plasmodium's proliferation creates toxic metabolic end products, which are released in the blood and cause the regular fever attacks that are typical of malaria.
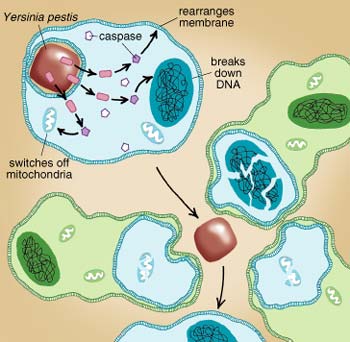
A few pathogens use a rather sophisticated approach to kill the infected cell: They trigger it to commit suicide. This process, called apoptosis, is not a sudden burst of the cell, but rather a controlled shutdown of the cell's biochemical machinery and disassembly of its constituents.
Apoptosis is not unique to infected cells, but is a normal process that can be triggered by a variety of stimuli, such as radiation or the stimulation of proteins on the cell's surface, such as the CD95 or the P2X7 receptors. Caspases, a family of enzymes that are specialized in cleaving proteins, play a key role in apoptosis. Once activated, caspases trigger further events in the cell that break down the DNA in the nucleus, switch off and disintegrate the mitochondria (the energy-producing cell organelles) and lead to a rearrangement of the surrounding plasma membrane. This chain of events finally causes the cell to shrink and decompose into smaller particles that are taken up and digested by macrophages in the vicinity.Shigella triggers apoptosis of the infected macrophage in order to gain access to the underlying tissue, from which it can spread to other cells. Also, the ability to drive macrophages to cell death can protect a pathogen from being killed by those cells. Yersinia, for example, induces apoptosis as a way to escape from a macrophage before being digested. In fact, Yersinia bacteria that are unable to trigger apoptosis because of a defective YopJ protein are unable to kill macrophages and are thus less virulent than Yersinia with an intact YopJ protein.
Bacteria may trigger apoptosis by injecting proteins via their type III secretion system into the target cell. For instance, some of the proteins that Shigella and Salmonella inject through the type III secretory system stimulate the host cell's caspase I proteins. Yersinia, the plague-causing bacterium, injects a protein called YopJ into the target cell to trigger its internalization. YopJ also inhibits certain signaling proteins in the host cell, which usually suppress apoptosis. The release of YopJ by the bacterium thus triggers suicide of the infected cell.
But apoptosis of the host cell may also be of benefit for the host, since pathogens released from the dying cell might then be taken up by macrophages that would destroy the pathogen. By sacrificing itself, the infected cell thus protects the body from a further progression of the infection. For instance, infection with Pseudomonas aeruginosa, an opportunistic bacterium that infects people with cystic fibrosis, triggers apoptosis of infected epithelial cells. Epithelial cells infected with Pseudomonas aeruginosa express two proteins on their surface: the CD95 receptor and the corresponding CD95 ligand. The ligand binds to the receptor and triggers apoptotic death of the infected cell. Indeed, mutant mice that are not able to express either the CD95 receptor or the ligand are killed by this pathogen, whereas mice that are able to express both proteins easily overcome a Pseudomonas aeruginosa infection. By analogy, the P2X7 receptor is assumed to play an important role in the killing of cells infected with Mycobacterium tuberculosis.
Bacteria are increasingly becoming resistant to various antibiotics. Multi-drug-resistant tuberculosis is growing into a serious health problem in Russia. For people with impaired immune systems, multi-resistant infectious bacteria have become a life-threatening problem. As the efficacy of existing drugs against many diseases declines, it is becoming increasingly important to find new ways to fight infectious organisms.
Moreover, there is yet no cure for most viral diseases. AIDS is devastating the population and economies of many African and Asian countries. At the same time, drug resistance is aiding the resurgence of malaria in Southeast Asia and Western Africa.
By understanding the nature of the infectious process, drug developers in academia and industry will gain the necessary knowledge to develop new treatments. These new approaches at fighting infections do not aim to kill the infecting pathogen as classical antibiotics do. If a pathogen can be prevented from entering and hiding in host cells, it will be visible to the immune system and thus killed by the body's own defense mechanisms. Pathogens are also less likely to evolve resistance to drugs that work this way. Treatments based on this concept include small molecules that block the surface structures that pathogens use to recognize and attach to target cells and block their ability to trigger internalization. The understanding of host-cell resistance can also open up new possibilities for treatment. A number of biotechnology companies have already developed anti-infectives and are testing their efficiency to prevent and cure bacterial infections in clinical trials.
The authors are indebted to their collaborators Heike Grassmé, Stephan Huber and Verena Jendrossek for sharing their skills and wisdom.
Click "American Scientist" to access home page
American Scientist Comments and Discussion
To discuss our articles or comment on them, please share them and tag American Scientist on social media platforms. Here are links to our profiles on Twitter, Facebook, and LinkedIn.
If we re-share your post, we will moderate comments/discussion following our comments policy.