Is Drug Addiction a Brain Disease?
By Marc Grifell, Carl L. Hart
This popular claim lacks evidence and leads to poor policy.
This popular claim lacks evidence and leads to poor policy.

The notion that drug addiction is a brain disease has become axiomatic. Around the globe aspiring health professionals treating substance abuse are indoctrinated with this belief, especially after the idea became popular in the 1990s. Its popularity extends far beyond the hallowed halls of academia. Both the May 1997 Time and the September 2017 National Geographic magazines were dedicated to the brain science of addiction. Numerous other popular magazines have run similar cover stories over the past two decades.
But after 20 years of research, one of us (Hart) saw that paradigm yielding dismal results. Meanwhile, behavioral research on outcomes after providing both animals and humans with attractive alternatives to drugs has yielded positive results regarding effective treatments, despite the lack of mainstream attention. This observation prompted Hart to refocus his research on these behavioral treatments. So in 2016 we teamed up to reexamine the prevailing assumptions supporting the brain-disease model of addiction and the data behind those assumptions. Like many other people in addiction research, coauthor Grifell had not directly questioned this paradigm until teaming up with Hart and digging into the evidence. Brain-imaging data from methamphetamine-addicted users provide the strongest support for the prevailing paradigm but still can be interpreted in other ways.

Brian Hubble
The concept of the addicted brain appeals to the intuitive idea that because recreational drugs exert their actions (for example, eliciting euphoria) through specific brain structures, there must exist discernible and meaningful brain differences between those who are afflicted with addiction and those who are not. These differences are then seen as the cause of the addiction and, therefore, as targets for treatment. The “diseased brain” perspective is appealing also because it offers treatment approaches that seem straightforward.
This view was consolidated and persuasively expressed in 1997 when the director of the U.S. National Institute on Drug Abuse (NIDA), Alan Leshner, published an influential editorial in the journal Science titled “Addiction is a brain disease, and it matters.” He explained, “That addiction is tied to changes in brain structure and function is what makes it, fundamentally, a brain disease.” Multiple proponents, including current NIDA Director, Nora Volkow, neuroscience researchers, addiction experts, and even politicians, have echoed these sentiments.
Despite this seemingly solid scientific consensus, there are virtually no data in humans indicating that addiction is a disease of the brain in the way that, for instance, Huntington’s or Parkinson’s are diseases of the brain. The existing paradigm is based on intuition and political necessity, not on data and useful clinical results. Yet the diseased-brain perspective has outsized influence on research funding and direction, as well as on how drug use and addiction are viewed around the globe. This situation contributes to unrealistic, costly, and harmful drug policies: If the problem is a person’s neurobiological state after exposure to a drug, then either the drug must be eradicated from society through law enforcement or an individual’s brain must be treated. In such a myopic approach, the socioeconomic and societal factors that contribute to drug addiction are considered a footnote in research, clinical practices, and policy, despite their apparent importance.
It’s easy to fall into circular thinking by defining addiction as a brain disorder and then researching it as a brain disorder. Knowing that someone uses a drug, even regularly, does not tell us whether that person is “addicted.” It doesn’t even mean that the person has a drug problem.
To meet the most widely accepted definition of addiction—the one in psychiatry’s Diagnostic and Statistical Manual of Mental Disorders, 5th Edition (DSM-5)—a person’s drug use must interfere with important life functions such as parenting, work, and intimate relationships. This use must continue despite ongoing negative consequences, taking up a great deal of time and mental energy and persisting in the face of repeated attempts to stop or cut back. It also may include the experience of needing more of the drug to get the same effect (tolerance) and suffering withdrawal symptoms if use suddenly ceases.
But 75 percent or more of drug users—whether they use alcohol, tobacco, prescription medications, or other drugs—do not meet criteria for drug addiction, according to numerous studies, including those by James C. Anthony of Michigan State University and his colleagues. Indeed, research shows repeatedly that such issues affect only 10 percent to 25 percent of those who use even the most stigmatized drugs, such as heroin, methamphetamine, and crack cocaine. Our use of the term addiction here is interchangeable with DSM-5’s substance use disorder, which always means problematic use of the sort that interferes with functioning—not just regularly ingesting a substance.
Even under the DSM-5’s more subtle definition of addiction, drug use could fit under the brain-disease theory. To investigate whether that is the case, we must look at the studies of substance abusers’ brains. The utility of any theory is measured by how well it can account for, predict, and even suggest strategies to control or treat specific phenomena. In Parkinson’s disease, for example, the dominant theory asserts that after the loss of more than 30 percent of dopamine neurons in the midbrain, clinical manifestation of the illness will commence. Although Parkinson’s disease progresses inexorably—that is to say, it is irreversible and fatal—the brain-disease theory used to explain the mechanism underlying the disease’s symptoms has led to the development of treatments that control the telltale gait, rigidity, and slow movement. Without dopamine replacement, currently considered the gold-standard treatment, advanced-stage Parkinson’s patients are unable to move.
Viewing this illness from a neurobiological perspective provides clear, indisputable evidence of the superiority of this approach compared with a psychological or behavioral theory. Such a theory might, for example, emphasize an environmental or behavioral modification to lessen Parkinsonian symptoms. This approach, of course, would be inappropriate and less effective than neuropharmacological manipulation consisting of dopamine replacement.
Barbara Aulicino
In the case of addiction, however, the diseased-brain theory does not have such clear explanatory power, especially when informing guidelines to treat the disorder. Behavioral and psychosocial therapies, such as cognitive behavioral therapy, contingency management, or motivational interviewing, remain the predominant treatments for substance-use disorders.
It is true that over the past several decades, data from basic research have contributed to an increased understanding of neural mechanisms involved in many effects produced by recreational drugs. Indeed, much of this evidence is the bedrock on which the brain-disease theory is built. But simply knowing that a drug causes, for instance, an increase in dopamine transmission does not necessarily provide any information about addiction to that drug.
We need to also make clear that specific neural elements underlying addiction have yet to be identified. Despite this empirical void, a key supposition of this theory purports that detectable brain dysfunctions in individuals afflicted with addiction are the source of the problem and that focusing on the brain should be paramount in our search for a solution.
Some of the most compelling evidence that seems to support the diseased-brain view of addiction comes from studies, mostly published in the 1980s and 1990s, of laboratory animals that were administered amphetamine. During these studies, the short- and long-term effects of amphetamines on neural structure and functioning as well as behavior were investigated. One of the most consistent findings is that a single large amphetamine dose administered to nontolerant animals produces extensive damage to dopamine neurons, meaning neurotoxicity. Areas of the brain that are rich in dopamine serve a wide range of important human functions, from mood regulation to movement to learning and memory. Indeed, a substantial database collected in laboratory animals indicates that large amphetamine doses produce disruptive effects in multiple behavioral domains, including learning and memory.
Because d-amphetamine and methamphetamine are used in several countries, including the United States, to treat a variety of disorders, such as attention-deficit hyperactivity disorder (ADHD), narcolepsy, and obesity, it is not difficult to see how the possibility of amphetamine-induced neurotoxicity might cause alarm. This information also raises concerns about the potential harmful consequences of methamphetamine addiction on the human brain and behavior.
Still, an important question remains: To what extent do the neurotoxicity data collected in animals extrapolate to people who may use amphetamine medically or who may be addicted to this class of drug? In other words, is this research relevant to humans who use amphetamines? Furthermore, amphetamine-induced neurotoxicity in animals can be prevented with previous exposure to several days of escalating amphetamine doses. Put more simply: Dosing regimens used in animal studies do not correspond with those typically used by humans; and the development of tolerance seen in long-term use can be protective against amphetamine neurotoxicity.
These caveats are critical because human recreational drug users—as well as patients on prescription amphetamines—usually increase (or decrease) their doses gradually as they gain more experience with a particular substance. This observation suggests that deleterious neurobiological and behavioral changes observed in many animal studies may not be apparent in human amphetamine users. It also highlights the importance of employing relevant models in future investigations of drug-related effects on brain functioning and behavior in animals.
Over the past two decades, research investigating the direct effects of amphetamine use on human functioning has steadily increased. The immediate effects of low to moderate doses of amphetamine or methamphetamine are clear and consistent: increased energy, enhanced ability to focus and concentrate, reduced subjective feelings of tiredness, and attenuation of cognitive disruptions brought about by fatigue or sleep deprivation. It is precisely because of these effects that several nations’ militaries, including the United States, have used (and continue to use) amphetamine in specific situations since World War II. Amphetamines help specialized professionals work better and longer in critical situations.
The impact of long-term administration of large drug doses in humans is less clear, owing to ethical considerations. An alternative approach to determining the extent to which large methamphetamine doses might produce brain or behavioral detrimental effects is to compare the brains and cognitive performance of healthy control participants with abstinent individuals whose drug use meets criteria for methamphetamine addiction. The idea is that regular use of illicit methamphetamine over several years may result in neurotoxic effects to specific neurons, including dopamine neurons. Damage to these neurons, of course, can have disruptive consequences on specific behaviors such as cognitive functioning and movement.
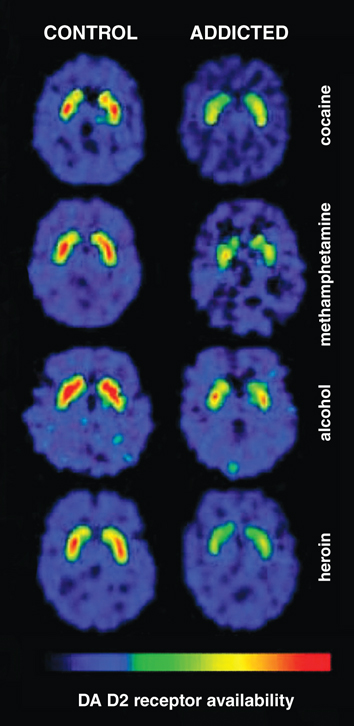
Typically, these studies combine brain-imaging techniques with cognitive testing so that brain structure integrity or activity can be correlated with a relevant behavior. Here, we will focus on studies using positron emission tomography (PET) imaging techniques for several reasons. First, PET studies provide the most consistent findings to date regarding neurobiological differences between individuals with substance use disorders and control participants. Second, PET imaging is currently the most suitable technique to detect dopamine transporter density and dopamine receptor availability. This function is relevant because an ample amount of evidence collected in laboratory animals demonstrates that large, repeated methamphetamine doses decrease these dopamine markers.
Science Source
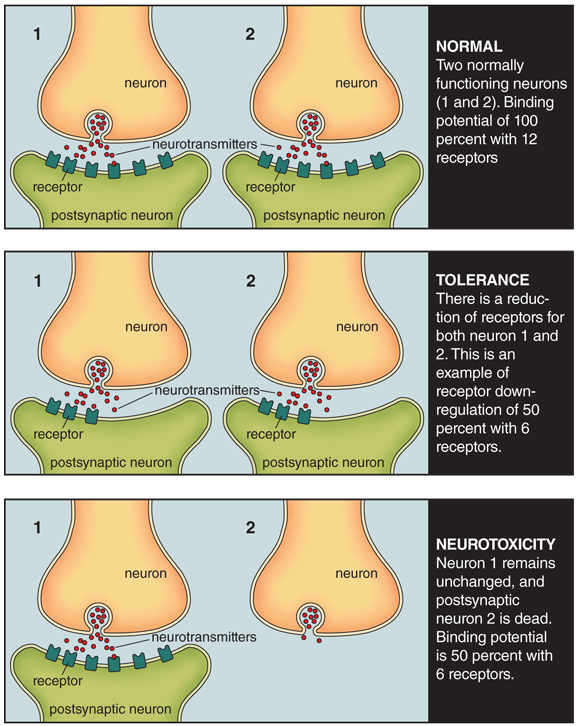
Consequently, some researchers have reasoned that long-term abuse of methamphetamine by humans should produce a reduction in dopamine transporter density and dopamine receptor availability. If this reasoning were correct, PET imaging studies would show these brain differences, which then may be interpreted as pathological changes in the brain produced by methamphetamine. Finally, this reasoning in turn would lead to the conclusion that such changes are the cause of addiction.
With PET techniques, a radioactively labeled chemical is injected into the bloodstream, and then a computerized scanning device maps out the relative amounts of the chemical in various brain regions. For our purposes, radioactively labeled drugs that bind to specific receptors or transporters are used, and so it is possible to see the extent to which binding occurs in the living human brain. In case of damage or death of dopamine neurons we would expect to see less binding of the radioactive dopamine-binding drug, because of lower availability of dopamine receptors.
In this way, PET provides an indirect measure of toxicity in the living human brain. This measure is indirect because neuron toxicity cannot be definitively determined in PET studies alone. Additional procedures or techniques are needed to definitively measure toxicity, because changes in radioactive binding may reflect adaptation (such as downregulation in tolerance development) of the neuron and not toxicity (see the two figures above).
Many comparative studies have been conducted over the past several decades that assess how well observations of methamphetamine’s effects in animals are consistent with those in people, but these comparisons are often inconclusive about cause and effect because of their necessary experimental design. In the human studies, methamphetamine users and healthy control participants are recruited. Then each individual undergoes a single brain scan and completes a cognitive task battery. An advantage of this approach is that it allows researchers to determine brain or cognitive differences between the groups at the time of testing. Brain images, however, are typically collected at only a single time point for both groups of participants.
This methodology makes it virtually impossible to determine whether methamphetamine use (or any other drug use) caused any observed differences, because preexisting differences between the two groups cannot be ruled out. Usually authors tend to adhere to the brain-disease model of addiction by attributing these differences to prolonged methamphetamine use, using words such as “changes,” “reductions,” “deterioration,” or “atrophy,” among others. This implication is clearly inappropriate, because these words imply a temporality that is not evaluated in these cross-sectional studies.
A caveat to the above is that a few recent studies have scanned participants’ brains over multiple time points. These studies have focused on how these neuromarkers differ between the methamphetamine users who are able to remain abstinent and those who keep consuming. Since these findings do not add new information in the comparison between methamphetamine users and healthy controls, we have focused primarily on studies when abstinent methamphetamine users were compared with control participants.
One consistent finding in such studies was lower striatal dopamine transport density in methamphetamine users compared with control participants. Because the striatum is a critical component of the brain’s circuitry related to reward and motivation, some have argued that this difference is indicative of a diseased brain following drug addiction, while others caution that neural differences do not necessarily equate to deficits or pathology. Because acute administration of methamphetamine seems to increase motivation through dopaminergic activity in the striatum, a reduction in its transport would be consistent with the development of tolerance to this particular effect.
Nearly 20 years ago, a study showed that taxi drivers in London had larger hippocampal volume than control participants who were not taxi drivers. No one would conclude, however, that either group was impaired or pathological.
One of the most rigorous studies, published in 2006, reporting dopamine transport density differences between the groups illustrates how specific neural data might be overinterpreted in these studies. Chris-Ellyn Johanson, now retired from Wayne State University, and her colleagues used PET imaging procedures and a comprehensive cognitive battery to assess brain functioning. The researchers found that dopamine binding potentials at a specific area of the brain were 10 percent to 15 percent lower in methamphetamine users.
There was considerable overlap, however, in the binding potentials of methamphetamine participants and those of the control group. As is shown in the figure below, monoamine binding potential values for some methamphetamine users were equal to or higher than those of some individuals in the control group, suggesting that the clinical relevance of the observed statistical differences might be limited.
In practical terms, that result means that if these brain images were shuffled into a single set, experts would not be able to distinguish between the brains of controls and those of abstinent users. Furthermore, if one subscribes to the brain-disease theory, one would anticipate that individuals with the largest methamphetamine addiction histories would have the lowest dopamine transporter binding potentials. This was not the case. Not only do imaging studies fail to show the expected drug-related changes in the brain, but clinical studies of addicts don’t show the expected functional changes, either.
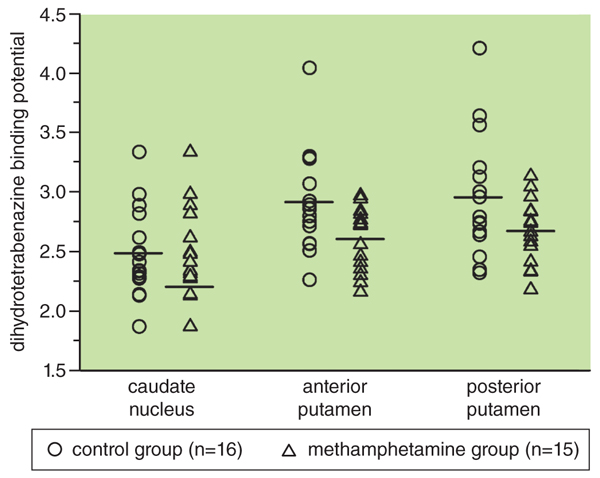
Graph adapted from Johanson, et al. Psychopharmacology 186:620. Springer-Verlag 2006
Some proponents of the brain-disease theory have implied that as more sensitive methodologies are developed, clear brain differences will be revealed. For example, in the supplement to her 2016 New England Journal of Medicine paper, Volkow argued that the “overlap [in binding potentials of drug users and healthy controls] is likely to reflect the limitation of currently available brain imaging techniques.” In other words, she suggests that our faith in future technologies will be reinforced by the demonstration of methamphetamine- or other drug-induced neurotoxicity. It seems, much to our chagrin, that some addiction-as-a-brain-disease scientists are encouraging faith-based thinking rather than applying evidence-based methods to inform drug addiction theories.
Nobel Prize winner and neuropathologist and -anatomist Santiago Ramón y Cajal perhaps stated it best when he observed in 1906: “Present-day science, in spite of its well-founded conclusions, has not the right to foretell the future. Our assertion can go no further than the revelations of contemporary methods.” The present data indicate that we do not have the evidence to support claims that the brains of addicted persons can be distinguished from those of nonaddicted individuals.
Regarding the cognitive findings in the study by Johanson and colleagues, the performance of methamphetamine users and control group participants did not significantly differ on most tasks. Methamphetamine users, however, performed more poorly than controls on measurements of sustained attention and immediate and long-term memory. Still, the methamphetamine users’ performance remained within the normal range for their age and educational group. In other words, the methamphetamine users were cognitively intact.
Although there are brain differences between addicts and nonaddicts, there are no data to support the idea that these differences qualify as a brain “disease” or even that some addictive drugs lead to a loss of brain function. Even though some drugs produce neurotoxicity when administered at high doses during long periods of time, it is not possible to generalize such effects to all drugs. To determine clinical relevance, cognitive scores should be compared against a normative database. Normative data, which are obtained from a large, randomly selected representative sample, incorporate important variables such as age and education, and establish a baseline distribution for a measurement. Unfortunately, this basic requirement is often ignored; as a result, the addiction literature is replete with a tendency to interpret any difference as deficits representing substantial loss of function.
This point is highlighted in the conclusions drawn from multiple studies that compare the cognitive or brain functioning of people with drug addiction with that of nonaddicted control participants. For example, Sara L. Simon and others at the University of California, Los Angeles, warned the following in a 2002 article in the Journal of Addictive Diseases:
“The national campaign against drugs should incorporate information about the cognitive deficits associated with methamphetamine….Law enforcement officers and treatment providers should be aware that impairments in memory and in the ability to manipulate information and change points of view (set) underlie comprehension….Methamphetamine abusers will not only have difficulty with inferences…but…they also may have comprehension deficits….The cognitive impairment associated with [methamphetamine abuse] should be publicized.”
Such warnings were based on measures that revealed statistically significant differences between methamphetamine users and controls, which alone are insufficient to determine true cognitive dysfunctions. Despite such inappropriately dire conclusions, findings from the bulk of the brain-imaging and cognitive literature assessing individuals who meet criteria for drug addiction indicate that they are virtually indistinguishable from their age- and education-matched non-drug-using counterparts. As methamphetamine neurotoxicity in animals has been the most widely demonstrated, the failure to replicate these results in humans leads us to conclude that it is a tremendous stretch to argue that the scientific data show that drug use causes brain disease.
Despite this empirical reality, the diseased-brain perspective has outsized influence on research funding and direction, as well as on how drug use and addiction are viewed in society. For example, the U.S. National Institutes of Health recently initiated a study that will cost taxpayers more than $300 million: The Adolescent Brain Cognitive Development longitudinal study primarily seeks to gather neuroimaging data to better understand the neural underpinnings of drug addiction among young people. Researchers will follow more than 10,000 people between the ages of 9 and 10 for a decade, collecting their genetic information and assessing their drug use and academic achievement.
This research endeavor will give less consideration to important social factors, such as parental income, neighborhood, or family structure. This oversight is to be expected, in part, because most of the lead investigators are neuroimaging researchers. Notably, there has never been such an ambitious funding effort focused on psychosocial determinants or consequences (for example, employment status, racial discrimination, neighborhood characteristics, or policing) of drug use or addiction.
The existing paradigm is based on intuition and political necessity, not on data and useful clinical results.
To be clear, our goal here is not to set up a dichotomy between biological and social factors involved in drug addiction, as if they are mutually exclusive. They are not. In addition, we recognize that many proponents of the diseased-brain theory of addiction habitually provide cursory and pro forma statements attesting to the importance of understanding the role of psychosocial and environmental factors in mediating drug addiction. These statements are often unconvincing because they are not accompanied with actions consistent with the claims.
Thus, our point is that there should be greater parity in the funding of drug addiction research and in how drug addiction is viewed. The weight of the evidence should drive research direction and funding. Through research published in 2016 that used the National Comorbidity Survey–Adolescent Supplement to study the substance use and mental disorders of more than 10,000 teenagers and also through our research, we now know that among the relatively small percentage of individuals who do become addicted, co-occurring psychiatric disorders and environmental and social factors account for a substantial proportion of these addictions.
This point indicates that far more research resources and efforts should be allocated to carefully studying these factors. It also suggests avenues for effective substance-use disorder interventions such as available mental healthcare and attractive alternatives such as career opportunities or sports facilities.
The argument for a more pluralistic view of drug addiction does not exclude a role for neuroscience, as long as data justify that role. At present, the utility of explaining drug addiction from an exclusive—or almost exclusive—diseased-brain perspective seems limited. A large proportion of people who previously were addicted managed to abstain without professional help, according to a couple of 2011 studies by Catalina Lopez-Quintero, Carlos Blanco, and their coauthors. For other people struggling with addiction, as noted above, the most effective treatments are behaviorally based.
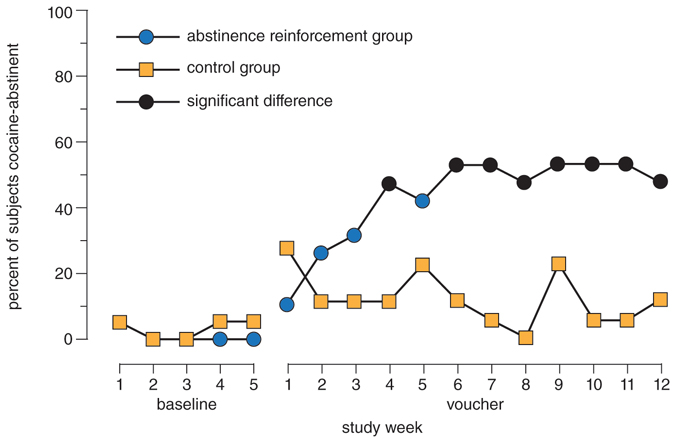
Higgins, S. T. 1997. The influence of alternate reinforcers on cocaine use and abuse: a brief review. Pharmacology Biochemistry and Behavior 57:419–427. Elsevier Science Inc.
Despite the effectiveness of evidenced-based behavioral therapies, they are not widely used, according to a 2016 review by Danielle Davis of University of Vermont and colleagues and a 2014 review by Kathleen M. Carroll of Yale University School of Medicine. Even when medication-assisted treatments are used, such treatments are far less effective without adjunctive behavioral therapies, as is argued in a 2000 paper in Drug Safety by James Bell and Deborah Zador of the Langton Centre in Australia.
Viewing addiction as a disease of the brain has demonstrated, thus far, limited utility for the development of effective strategies to deal with drug addiction. Notably, the most relevant biological treatments, such as methadone, disulfiram, and others, were developed prior to the establishment of the brain-disease model of addiction.
Disproportionately viewing drug addiction through the brain-disease lens contributes to unrealistic, costly, and harmful drug policies. If the real problem with drug addiction, for example, is the interaction between the drug itself and an individual’s brain, then the solution to this problem lies in one of two approaches: Either remove the drug from society through policies and law enforcement (for example, drug-free societies), or focus exclusively on the “addicted” individual’s brain as the problem. In both cases, there is neither genuine need for nor interest in understanding the role of socioeconomic factors in maintaining drug use or mediating drug addiction.
The entire removal of recreational psychoactive substances from society is both impractical and impossible. There has never in history been a drug-free society, and it is unlikely that there ever will be one. In spite of this fact, law enforcement is charged with the unenviable task of carrying out repressive recreational drug-use policies that emphasize abstinence. Despite the claim that viewing addiction as a brain disease would lessen stigma and reduce drug-related arrests, millions of people are arrested annually for drug possession. In the United States, for example, 2016 data from the U.S. Federal Bureau of Investigation indicate there were 1.5 million annual drug arrests, a number that hasn’t appreciably changed since 1996.
Even worse, the abhorrent practice of racism continues to flourish in drug law enforcement. In the United States, blacks are four times more likely than whites to be arrested for cannabis possession, even though both groups use cannabis at similar rates. And more than 80 percent of those convicted of heroin trafficking are black or Latino. There are considerably more white heroin users than black or Latino users, and most drug users buy their drugs from dealers within their own racial group.
An insidious assumption of the diseased-brain theory is that any use of certain drugs (for example, crack cocaine, heroin, or methamphetamine) is considered pathological, even the nonproblematic, recreational use that characterizes the experiences of the overwhelming majority who partake of these drugs. For example, in the 1980s crack cocaine addiction was said to occur after only one hit. Drug experts with neuroscience leanings weighed in. “The best way to reduce demand,” Yale University psychiatry professor Frank Gawin told Newsweek in 1986, “would be to have God redesign the human brain to change the way cocaine reacts with certain neurons.” For a more recent example, a popular U.S. antidrug campaign implies that one hit of methamphetamine is enough to cause irrevocable brain damage (http://www.methproject.org/ads/tv/deep-end.html).

Brian Hubble
“Neuro” remarks made about drugs, even though they have no foundation in evidence, can be pernicious: They can help shape an environment in which there is an unwarranted and unrealistic goal of eliminating certain types of drug use at any cost to marginalized citizens. In 1986, the U.S. Congress passed legislation setting penalties that were 100 times harsher for crack than for powder cocaine violations. More than 80 percent of those sentenced for crack cocaine offenses are black, despite the fact that the majority of the drug’s users are white.
Even today, some politicians enact misguided drug policy based on these neuroexaggerations. The recent actions of Philippine President Rodrigo Duterte represent but one example. A year into his presidency, more than 4,000 people accused of using or selling illegal drugs have been killed. This tragedy results from Duterte’s view of methamphetamine’s effects on the brain. Duterte justifies his actions by stating that methamphetamine shrinks the brains of users, and as a result, these individuals are no longer capable of rehabilitation. It is not difficult to see how Duterte’s misguided belief that methamphetamine causes brain damage might have been shaped by exaggerated claims drawn by researchers who tout that addiction is a brain disease.
Many addiction researchers begin with the assumption that this condition is a brain disease. Yet there are virtually no data in humans indicating that addiction is a brain disease in the way that, for instance, Huntington’s or Parkinson’s are brain diseases. The present evidence indicates that this assumption should be reevaluated to formulate a more accurate view of drug addiction. An evidence-informed view would be more inclusive, would emphasize a prominent role for psychosocial and environmental factors, and would focus on offering alternative reinforcers—nondrug alternatives that decrease problematic drug-taking. From a practical or clinical perspective, this approach means it is unacceptable to tell substance-use disorder patients that they suffer from a diseased brain. Instead, a comprehensive psychosocial assessment should be employed, and the resulting findings should dictate intervention strategies.
Research has now shown repeatedly that alternative reinforcers can be used effectively to treat substance-use disorders. This kind of treatment is called contingency management. The idea comes from basic behaviorism: Our actions are governed to a large extent by what we are rewarded for in our environment. These cause-and-effect relationships, where a reward is dependent (contingent) upon the person either doing or (in the case of drugs) not doing a particular behavior, can be used to help change all types of habits.
A body of literature now shows that providing alternative reinforcers improves addiction treatment outcomes. It is far more effective than using punitive measures such as incarceration, which often is less useful in the long run. Although many people stop or at least reduce their drug use while incarcerated, jail and prison don’t provide positive alternatives to replace drug habits. When heavy drug users return to their communities, they are not better equipped to find work and support themselves and their families; instead, having a criminal record and a gap in their résumé makes finding work even harder.
A 1993 study led by Stephen Higgins of the University of Vermont randomly assigned treatment-seeking cocaine users to either contingency management plus behavioral counseling or to a traditional 12-step-focused counseling treatment. Patients in the contingency-management arm of the study received vouchers for merchandise whenever they had drug-free urine. Fifty-eight percent of participants in the contingency-management group completed the 24-week outpatient treatment—compared with 11 percent in the 12-step group. In terms of abstinence, 68 percent in the contingency-management group achieved at least eight weeks cocaine-free, versus just 11 percent in the 12-step group. And after the rewards are stopped, people in contingency management are no more likely to relapse than other treatment graduates. Because more people complete treatment with contingency management, this outcome makes for an overall reduction in relapse.
More than three dozen studies have now been conducted on contingency management—used in the treatment of opioid, cocaine, alcohol, and multiple-drug addiction. They show that contingency management typically does better than treatment without it—and that larger, faster rewards are more effective than smaller, less quickly received incentives. This result is exactly what research on other types of behavior predicts.
Recently, we have asked our colleagues who are proponents of the brain-disease model of addiction to provide us with practical information that might be helpful to patients. Invariably, we are directed toward the future with promises of new and effective therapies. Multiple purported therapies (including the cocaine vaccine, anticraving medications, genetic therapy, and many others) have proven unsuccessful over the past two decades. In general, the response to such failure has not been to question the theory guiding the search for treatments, but instead to double down, making the theory even more complex. Meanwhile, addicted patients continue to suffer.
It is incumbent on us all to be on guard against neuroexaggeration and neurocentrism, because they have inappropriately and disproportionately influenced our research funding priorities and drug policies. The stakes are too high, and the human cost is incalculable.
Click "American Scientist" to access home page
American Scientist Comments and Discussion
To discuss our articles or comment on them, please share them and tag American Scientist on social media platforms. Here are links to our profiles on Twitter, Facebook, and LinkedIn.
If we re-share your post, we will moderate comments/discussion following our comments policy.