
This Article From Issue
November-December 2014
Volume 102, Number 6
Page 409
DOI: 10.1511/2014.111.409
In this roundup, associate editor Katie L. Burke summarizes notable recent developments in scientific research, selected from reports compiled in the free electronic newsletter Sigma Xi SmartBrief. Online: https://www.smartbrief.com/sigmaxi/index.jsp
Earth’s New Supercluster Home
By measuring the velocities of 8,000 galaxies, corrected for cosmic expansion, a group of astronomers have made the best measurement of the supercluster of galaxies that dominates our region of the cosmos. The researchers, led by Brent Tully of University of Hawaii, tracked galaxies that are flowing in the same direction, much as a watershed includes all water flowing into the same tributary. Such flows offer a novel way to infer the presence of matter by its gravitational pull, whether or not it is visible. Earth and the Milky Way were previously considered part of the Virgo supercluster, which now appears to be just one region of a much greater structure called Laniakea—“immeasurable heaven” in Hawaiian. The Laniakea supercluster measures 500 million light-years across and contains the mass of about 100 quadrillion suns. This work is part of an ongoing conversation in cosmology on how to map and define the large-scale structure of the universe.
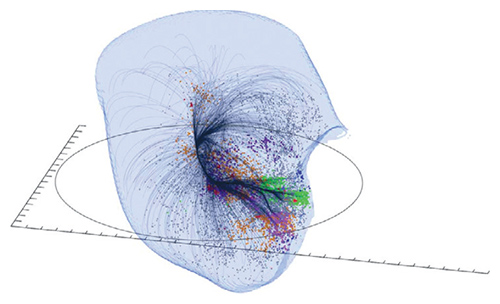
SDvision interactive visualization software by DP at CEA/Saclay, France
Tully, R. B., H. Courtois, Y. Hoffman, and D. Pomarède. The Laniakea supercluster of galaxies. Nature 513:71–73 (Published online September 3)
Reprogramming Stem Cells
A genome-wide screen yielded information about the barriers to effective cellular reprogramming, used to generate induced pluripotent stem cells (iPSCs), body cells that have been genetically regulated to act like undifferentiated stem cells. These stem cells hold great promise for regenerative medicine. However, iPSCs do not act completely like true embryonic stem cells, indicating that they hold onto bits of their somatic past. The process of making iPSCs is inefficient, too, because only a few viable iPSCs will emerge from the reprogramming of many somatic cells. The 956 genes identified inhibit reprogramming by many different mechanisms, and they may also be genes that, when they malfunction in vivo, could be associated with cancer. This comprehensive study will spawn new developments to increase the efficiency of iPSC generation.
Qin, H. Systematic identification of barriers to human iPSC generation. Cell 158:449–461 (July 17)
Bird Migration Evolved in North
Conventional wisdom in biology was that migratory birds mostly evolved from tropical birds that left to escape competition, but a new study indicates that most migratory birds actually have ancestors from temperate, not tropical, regions. Studying 823 species of American songbirds, University of Chicago’s Benjamin Winger and his colleagues found that intercontinental migration was twice as likely to evolve in birds with ancestors in North America than in those with neotropical ancestors, suggesting that birds migrated to escape harsh winters rather than to escape competition. Although migration could arise in temperate or tropical species, this new study showed it was much more likely in the former. The question remains whether this pattern holds for other bird lineages, and how changing climate could affect evolution in migratory birds.

Wikimedia Commons
Winger, B. M., F. K. Barker, and R. H. Ree. Temperate origins of long-distance seasonal migration in New World songbirds. Proceedings of the National Academy of Sciences of the U.S.A. 111:12115–12120 (August 19)
Neanderthals Died Out Earlier
A new study using improved radiocarbon dating techniques suggests that Neanderthals in Europe died out about 39,000 years ago—more than 10,000 years earlier than originally thought. This time period corresponds with the spread of anatomically modern humans across Europe, adding support to the contention that the presence of modern humans was detrimental to Neanderthal populations. Although there is not evidence to suggest that the two populations killed one another outright, modern humans and Neanderthals may have competed for space or food. This study adds fuel to the debate over when and how Neanderthals died out.
Higham, T., et al. The timing and spatiotemporal patterning of Neanderthal disappearance. Nature 512: 306–309 (August 21)
Insulin Inert in Hibernating Bears
Studying hibernation could lead to better treatments for type 2 diabetes: New research shows that hibernating grizzly bears enter a state in which insulin, which prevents breakdown of body fat, stops working. Lynne Nelson of Washington Sate University and several colleagues sampled tissue from six captive grizzly bears for one year, monitoring insulin activity. They found that insulin activity was normal—by human standards—in all seasons, except the winter months when the bears are inactive and do not eat, drink, or defecate. During this time, the animals show the same symptoms we associate with type 2 diabetes: high fat levels and insulin resistance. This process allows the bears to break down fat stores while they are in such an inactive state. Understanding the chemical pathway that alters insulin sensitivity in bears could have applications in human therapies.

Wikimedia Commons
Nelson, O. L., et al. Grizzly bears exhibit augmented insulin sensitivity while obese prior to a reversible insulin resistance during hibernation. Cell Metabolism 20:376–382 (August 5)

American Scientist Comments and Discussion
To discuss our articles or comment on them, please share them and tag American Scientist on social media platforms. Here are links to our profiles on Twitter, Facebook, and LinkedIn.
If we re-share your post, we will moderate comments/discussion following our comments policy.