The Origins of Lying and Deception in Everyday Life
By Michael Lewis
How do children make sense of the complex social code that dictates when they should or should not lie?
How do children make sense of the complex social code that dictates when they should or should not lie?

DOI: 10.1511/2015.113.128
Those of us living in the United States seem particularly concerned about lying and deception. Consider our myth about our founding father and general of our Continental Army, George Washington. What most of us know best about Washington as a child—in fact, the only thing we know—is that when asked by his father, “Did you chop down the cherry tree?” he is reported to have told the truth. Whether the story is accurate or not (it was likely made up by biographer Parson Weems), the fact that children are taught the first president of our nation never lied speaks to the importance of the issue for American parents.

Given the diversity of the studies on this topic, it may be helpful to consider what we mean by lying and deception, both in children and in adults, and to use these definitions to create a taxonomy that can inform our ideas about the origins of lying and deception. While the topic of lying and deception in emotional life has received some attention (see, for example, the works of Paul Ekman), there has been relatively little investigation of this behavior in children. Apart from Charles Darwin’s description of his child at two years old and a single study published by philosopher Charles Hartshorne in 1928, it was not until the 1980s that research into lying and deception began in earnest, with the publication of studies by Carolyn Saarni and Pamela Cole as well as my own. Given the perennial interest in emotional development, it is somewhat surprising that this line of inquiry emerged so late. As we will see, the research demonstrates that facial expression can mask an individual’s inner emotional experience, even in children; moreover, it suggests that lying and deception, as measured by both verbal and facial expression, may have been subjected to evolutionary pressure and are positively related to other cognitive capacities associated with psychological fitness.
On its face, it is obvious that the notion of something being truthful implies that there also can exist falseness and lies. Our human experience confirms that lying, falsehood, and the masking of our inner selves exist as part of the social world in which we live. Saarni, a professor of psychology at Sonoma University, has shown some of the methods people use to deceive, which derive from social norms prescribing how people should express their feelings. Such rules of comportment usually seem completely natural to the people who have been brought up with them, yet they vary greatly from one culture to another. In Japan, for example, girls more often than boys are taught to hold their hand in front of their mouth when they laugh, and men as well as women are taught to suppress facial expressions of anger. In another example of differing cultural and personal norms for the display of emotion, Marsha Weintraub, of Temple University, and I observed how two-year-olds react to their mothers’ departure: American children are often encouraged to express sadness at the time of parting, even though they quickly recover once their mothers are gone and they are left with the babysitter. We have found, too, that parents differ in their response to a toddler’s minor injuries, with some making soothing remarks like, “It’s okay to cry, it must really hurt,” whereas other parents reproach their toddlers (especially boys) for crying. In a real sense, deception, lying, and dissemblance are indeed a “natural” feature of the social environment.
Of course, there are many varieties and degrees of deception; masking one’s emotions is very different from deliberately uttering a falsehood. One way to sort out the various forms of lying is to consider the state of the deceiver’s awareness of his or her deception.
When children tell their parents that they have done their chores even though they have not, or when students claim they could not finish their assignment on time because they had to help a sick friend, these types of lies are self-aware. The deceiver knows what was expected of him or her and seeks to hide the failure. Deceptions of this type more often than not are subject to societal scorn, which we express with phrases such as, “You ought to be brave enough to take your punishment.”
Deception or lying without awareness is more problematic. If someone makes a false statement without knowing that what she or he said is contrary to fact, the speaker cannot be said to be lying. Acknowledging this point then brings us to questions about deception in nonhuman animals. Although there is considerable evidence of deceitful behavior in animals, it is not clear that chimpanzees, for example, have a third-level perspective that can be characterized as “I know the other chimp knows that I know.” Without this perspective, animal deception has to be distinct from that of humans.
In earlier work, I have suggested a taxonomy of lying and deception that may be useful in examining children’s behavior in this regard. This taxonomy may turn out not to encompass all types of deception, nor does it imply that the types are mutually exclusive. In many cases, an act of deception may meet more than one of these criteria:
Lying to protect the feelings of another;
Lying for self-protection to avoid punishment;
Lying to the self, or self-deception; and
Lying to hurt others.
The first three types, although often considered moral imperfections, will be shown to be positively related to other cognitive skills.
Rana, a three-year-old girl, was eagerly awaiting a Christmas present from her grandmother. She’s hoping for a fun toy. Nevertheless, when her grandmother presents her with a sweater she has knitted, Rana rips open the package and smiles at her grandmother, saying, “I like it.”
Rana, like many children her age, has already learned how to adjust her facial expression and speech to meet social requirements. This practice is often taught under the name of “little white lies,” although I prefer the description “lying to protect the feelings of others.” In The Rise of Consciousness and the Development of Emotional Life, I have argued that the function of this type of deception is socially adaptive. As summarized by the Norwegian sociologist Stein Braten, there is considerable evidence that the human infant has built-in mechanisms for helping others. Although some say that such deception detracts from interpersonal relationships, it seems reasonable to assume that the maintenance of social interaction requires some deceptions of this type.

Illustration by Tom Dunne.
Everyday examples of this motive abound, but studying it in young children in the laboratory poses some difficulties. Saarni, who studies how children develop emotional competence, has reasoned that if a child was promised an attractive toy but did not receive it, the child might be unlikely to show her disappointment so as not to make the experimenter feel bad. In her studies, children aged seven to eleven were given the choice of several toys and asked which one they liked best and which one they liked least. The children were then given a problem to solve, with the promise that they would receive the toy they liked best. After solving the problem, each child was given his or her least-liked toy. In the expression on their faces and in their spoken remarks, the children’s responses differed both by sex and by age. Younger children, especially boys, were likely to show more disappointment, whereas older children showed more positive emotions. In a similar study at the University of Pennsylvania, this time using children as young as four years, Cole, too, showed that girls were better at hiding their disappointment than boys; in this case, however, the four-year-olds were as good as older children in masking their expression of sadness. When this study was extended to preschoolers, three-year-olds also inhibited their disappointment. Clearly, this ability makes its appearance quite early in life.

Illustration by Tom Dunne.
Other paradigms have been used to test whether and when young children lie to protect the feelings of another. One such study is a variant of the classic Rouge test, which I developed in 1979 together with my then-graduate student Jeanne Brooks-Gunn: we put a red mark on a child’s nose and placed him in front of a mirror. We found that by the age of two, most children touch their noses and show embarrassment when they see the red spot in the mirror, a reaction suggesting they understand that this mark is unusual and out of place. In a variation of this test, Kang Lee and his associates created another paradigm to examine the problem of lying to protect the feelings of another. Here the experimenter, who this time has a red mark on her own nose, tells the child she is about to have her picture taken and asks whether she looks okay. After the experimenter (E1) has her picture taken and leaves the room, another experimenter (E2) enters and asks if experimenter E1 looked okay.
In this study of 98 children between three and seven years old, 89 percent lied, saying that experimenter E1 looked okay. Interestingly, the 11 percent who told the truth, saying the experimenter did not look okay, showed facial expressions quite similar to those who lied. Moreover, there were no age effects. From such data, it seems that from the ages of three to seven, children are more or less equally skilled at polite deception.
Often, their early coaching in this skill takes place in the home. Given that parents lie to spare the feelings of others, they are likely to coach their children in this practice (“Tell Grandma that you like her gift/cooking/choice of movie even though it may not be what you wanted”). Children may also see their parents engage in deception to save the feelings of others. Thus, for example, Rana may hear her mother say, “My friend is coming over for tea and I am too tired to see her,” but when the friend arrives, the child sees her mother smiling and hears her say, “I am so glad you stopped by.”
In looking at the effect of adults’ telling the children to lie to spare the feelings of another, child psychologists Victoria Talwar and Kang Lee, then at Queen’s University in Ontario, used the “disappointment” paradigm developed by Saarni and Cole, but with the addition that adults encouraged children to tell a white lie. The majority of the children, even by three years old, complied; when disappointed, girls smiled more often than boys, an observation that suggests they were better able to mask their feelings as required.
Although the experimental data are still limited, they show that deception to spare the feelings of others can be seen as early as three years of age and that girls may be better at it than boys. Moreover, parental instructions to lie to protect the feelings of another appear to be effective.
These studies touch on the interesting challenge between a child’s developing moral behavior and his or her developing prosocial behavior—that is, any behavior that is intended to help others. Clearly there is a conflict between not telling a lie and not hurting another’s feelings, especially given that honesty is considered to be part of moral behavior. To address this question, in 2009 Gail Heyman led a study at the University of California–San Diego in which children were asked their reasons for telling a lie. The researchers gave children between the ages of seven and 12 years old a series of vignettes, some of which were about children who receive an undesirable gift and are asked whether they like it or not. In half the stories the children tell the truth, and in the other half they do not. In another series of stories, a child transgresses by damaging a library book. Again, in half the stories the children confess to the damage, whereas in the other half they do not.
When asked to evaluate the behavior of the children in the stories they had been told, the participants in this study gave more neutral ratings of the politeness stories and more negative ratings of the transgression stories. Such findings as these support the view that children learn and evaluate positively lies that protect the feelings of others more than other types of lies.
Given the growing body of literature (as summarized by Braten) suggesting that toddlers demonstrate early prosocial behavior, lying to spare someone’s feelings is another aspect of this behavior. More support for the prosocial view of lying comes from an unpublished study by developmental psychologist Antonella Brighi, of the University of Bologna, who found that four-year-olds who masked their emotions more successfully when placed in the “disappointing gift” paradigm were more likely to be chosen by other children to be their play dates. As we will see, lying and deception are often associated with other prosocial and cognitive abilities.
Two-year-old Maron is told not to eat a cookie, but when his mother is out of the kitchen, he does so. When his mother questions him about eating the cookie, he lies and says “No.”
Another common form of lying in children has to do with the motive to avoid punishment. Children learn readily to lie when they have committed some undesirable act or have not done something they were asked to do. In this example, Maron remembers that eating a cookie has in the past evoked parental anger or punishment, and he tries to avoid these predictable consequences by lying.

Photographs courtesy of Michael Lewis.
Once toddlers have grasped the concept that lying may offer a way of escaping punishment, what do they do? To study this type of deception in children too young to speak fluently, my colleagues and I needed an experimental situation in which very little verbal behavior was required; our creative solution was the “do not peek” paradigm. In these studies, a young child is brought into a room and seated at a table, facing forward. Behind the child’s back, the experimenter unpacks an elaborate toy, saying, “Do not turn around and look at the toy.” After the toy is set up, the experimenter tells the child that she must leave the room for a few minutes. Once again, as the experimenter leaves the room, the child is told, “Do not peek at the toy, we will play with it when I come back.”
We designed this situation to maximize the likelihood that the child will peek, and indeed this is often the case. The children are left alone in the room for a short time, and if after five minutes they still have not peeked, the experimenter returns. The experimenter asks the child “Did you peek?” Unknown to the children (but with the consent of their parents), they are videotaped throughout the experiment to provide an objective record of whether they actually peeked and are now lying or telling the truth.
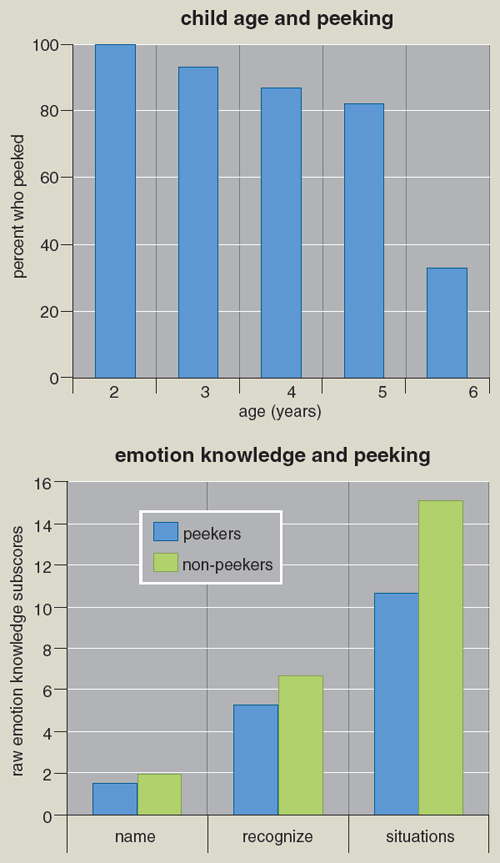
The results of our first study indicated that children as young as two and a half are capable of deception. Most children this age will violate the “no-peeking” rule if left alone; only four of the 33 subjects were able to refrain for the full five minutes. Of those who peeked, 38 percent admitted to peeking, 38 percent denied it, and 24 percent gave no verbal response. Thus, 62 percent of our subjects between the ages of two and a half and three deceived in some way. Facial and bodily activity did not differentiate the deceivers from the truth-tellers, suggesting these youngsters had already learned to lie fairly successfully.
Tom Dunne.
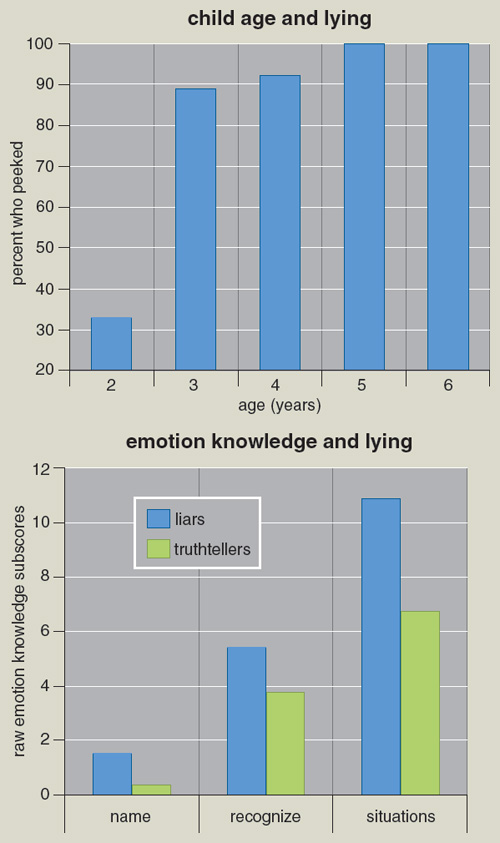
In a similar study, we looked at more than 180 children between three and six years of age. As expected from other studies, as children become older, they become better at resisting temptation. By the age of six, 35 percent of the children were able to sit with nothing to do and resist the temptation to peek. At the same time the prevalence of lying increased, so that by at age six all the children who had peeked denied having done so. While 25 percent of children two to three years of age admit to peeking, this number drops to near zero when the children are five years or older. Thus, even by two to three years, most children have learned to lie when they violate a rule. Although girls are less likely to peek than boys, there are no sex differences in lying.
From the similar data on age-related changes in lying that have been found in Japanese, West African, and Chinese children, it appears that lying to avoid punishment may well be a universal phenomenon. Moreover, lying to avoid punishment becomes more common as the child grows older, a pattern that also has been repeatedly observed. Given the widespread use of this type of lie, as well as its increase with the children’s increasing age, it may be an important adaptive response in humans, associated with other adaptive functions and competencies. Telling a lie successfully requires not only the ability to create a false belief deliberately but also to have some idea about what another person may or may not know; this capacity, known as a “theory of mind,” commonly develops in children around the age of two to three years.
The “peeking task” itself taps two skills: the ability to delay or avoid peeking, and once having peeked, the ability to lie. Although it is not central to deception, a child’s inability to resist peeking is related to several features associated with dysfunction. As an example, the speed with which a child gives in to the temptation to peek is inversely related to his or her IQ and emotional intelligence: Those who peek sooner tend to have lower scores on emotional knowledge as measured by tests such as being able to name emotional faces when shown to them, and lower scores on their knowledge about what emotions are likely to be seen in particular contexts. Children from risky family environments peek sooner, as do children with higher neonatal risk scores. The ability to resist peeking for a shorter or longer interval is clearly related to children’s competence.
However, the picture changes when lying is examined with regard to the child’s social and cognitive competence. Children with higher IQ are more likely to lie than those with lower IQ. Moreover, children who score higher on measures of emotional knowledge are also more likely to lie than truth-tellers. The truth-tellers had lower IQ scores by more than 10 points.
Tom Dunne.
Other studies of children from three to eight years old have looked at lying and its possible relation to various aspects of mental development. In another study by Talwar and Lee, children were asked about the nature of the toy after they denied having seen it. Younger children were unable not to name the toy, thus revealing they had peeked, whereas older children had no difficulty concealing the fact. In another study, children who lied and those who did not were compared on several tasks that assessed moral judgment, theory of mind, and executive functioning, which included the challenge of inhibiting certain responses. In all these assessments, children who had lied scored better than those who had told the truth—a result that strongly suggests the ability to lie is positively related to cognitive competencies!
Such findings support the view that people who commit a transgression and confess are less capable in many capacities, a view that bears important personal as well as sociobiological implications. Robert Trivers, in his book The Folly of Fools: The Logic of Deceit and Self-Deception in Everyday Life, argues that deception can be useful in protecting the immune system by reducing the stress response (that is, preventing an increase of the stress-related steroid cortisol) that would result from the child’s failure to meet her or his standards or goals. Increases in cortisol have been shown to be inversely related to immunological competence.
The notion that lying to protect oneself from punishment may be adaptive is consistent with the work of anthropologists such as Richard Byrne, who in 2004 found a positive relation between neocortex size and deception in primates. At the same time, the association of lying with prosocial behavior has been amply demonstrated. Moreover, psychologist Roy Baumeister and others have suggested that in some cases lying may be important for mental health, whereas Francesca Gino and others have shown lying to be related to creativity.
Any statement about the social benefits of lying would have to start from the premise that most individuals could easily master this skill at an early age. How good is young children’s ability to deceive? It may be better than we think.
Psychologists like Carroll Izard and Paul Ekman, who study facial expressions, have argued that the face does not lie; careful measurement of facial and bodily expressions, they claim, can always reveal deception. The work of other research groups, however, indicates that most people’s detection of lying is extremely poor. Angela Crossman and I looked at adults’ ability to spot children who were lying in the “do not peek” situation. When over 60 men and women watched video segments of 50 or more children saying they had not peeked—with some of the children telling the truth and others lying—adults did no better than chance, indicating they could not detect the liars. Videotape segments showing older children yielded similar results.
Studies of how children begin lying to protect themselves from punishment are more numerous than those of children lying in order to spare the feelings of others, and the findings are more robust. As discussed earlier, multiple studies indicate that lying crops up early in life, by two to three years of age, and that it increases as the child gets older. The most intriguing insight may come from the close association between a child’s ability to lie and his or her psychosocial competence, which in turn suggests that lying is somehow an adaptive behavior. This interpretation seems reasonable enough: Surely the wish to avoid punishment or harm is an adaptive trait.
The third type of deception in our taxonomy has been the hardest to study, particularly in young children. Nevertheless it is common among both adults and children, and clearly it presents both advantages and disadvantages:
Benjamin, a shy young man, calls a woman for a date and is told that she cannot see him because she is busy for the next three weekends. He now has a choice: He can conclude that she does not want to go out with him, and feel humiliated and shamed at the rejection. Alternatively, he can conclude he does not want to date such a busy woman. This spares him the shame and humiliation. In fact, both thoughts pass through his mind, but he remembers only that he does not want to date her.
The psychic advantage of this way of thinking seems clear; in some circumstances, there may be little reason to lower one’s self-esteem by being honest with oneself. On the other hand, self-deception may close off the possibility of learning from one’s mistakes or taking some necessary action—for example, if a person examines his body and discovers an unexplained lump. If the individual persuades himself that the lump has always been there—a false memory—he is likely to take no action. Should the lump be recognized later as a first sign of cancer, the delay of treatment stemming from this self-deception could bring serious consequences. It is fair to say self-deception may be psychologically valuable but also sometimes self-defeating.
Self-deception in children has received scant scientific attention to date, but fortunately there are data available on the development of pretend play, with which it shares many features. Self-deception takes the form of knowing X, and not knowing X, at the same time. Pretend play also has this form, because the child must hold in her mind the two opposite thoughts that the object of play X is not X. This view was suggested first by Jean Piaget and more recently by Alan Leslie, director of the Cognitive Development Laboratory at Rutgers, who also sees pretend play as involving double knowledge that requires the toddler’s ability to distinguish between what is real and what is not. The very fact that the child knows her action is not literal—“X is not X”—means that she has self-awareness. In fact, the studies I have conducted with 15- to 24-month-olds demonstrate that pretend play is related to the use of personal pronouns such as me or mine, and to mirror self-recognition, which, like pretend play, is typically present in children by age two.

Illustration by Tom Dunne.
Pretend play begins at about one year, when infants try to imitate the behavior of people around them. Psychologist Douglas Ramsay and I have suggested that in this type of pretense—for example, when an infant sees his mother talking into a phone and is encouraged to imitate her actions with his toy phone—the child is both the subject and the object. By age two or three, this behavior is gradually replaced by a more complex form in which the object is another person—for instance, a child might pretend that her doll is talking on the phone, rather than simply that she herself is talking on the phone. This form involves the pretense of both the imaginary phone and the doll talking on it.
By three years of age, a child’s ability to maintain internal rules and goals enables him to consider the success or failure of his behavior and even to apportion blame or credit for it. (Psychologist Carol Dweck gives an excellent account of this with older children.) This new ability also allows for the emergence of the self-conscious emotions of embarrassment, shame, guilt, and pride. In my book Shame: The Exposed Self, I show that by age three, children show shame when they fail a task and pride when they succeed. These powerful emotions provide the motivation for ways of thinking about themselves, which also allows them to self-deceive around their own success and failure.
Although it is easiest to observe in young children, where it may be dismissed as simple “play,” self-deception is important for emotional life at all ages. Moreover, as we and Trivers believe, self-deception may be needed for all forms of deception.
The fourth category in our taxonomy is the type of lie designed to inflict suffering. Far from being adaptive, such lies represent some form of psychopathology. For the most part, lying that injures another has not received much attention, although Richard Rogers has studied pathological lying. Of all of the four types of lies mentioned here, this is the least prosocial.

Illustration by Tom Dunne.
Attempting to protect oneself from punishment by lying about another is likewise maladaptive, because such lies, although sparing oneself from punishment, simply transfer the punishment to another person. From the point of view of a young child who scribbles on the wall, blaming the mischief on a sibling makes sense as a way to try to escape punishment. From a broader perspective, however, placing the blame on another is not an adaptive response, for it does not lead to social coherence.
Our concern about lying is not solely a national issue, even though we have as a focus the invented story of George Washington’s immaculately truthful childhood. Some studies with children across the globe suggest that lying and deception may exist as a feature of the human condition. Our current national debate about federal or state agencies lying to us is just part of the larger discussion in which the morality of lying is pitted against its evolutionary function and its prosocial needs.
Lying to others is most often seen as an interpersonal failure because it damages trust, believed to be one of the hallmarks of a relationship. Yet, as we have already noted, lying to protect the feelings of another appears a necessary act similar to other prosocial behaviors such as helping and empathy. Whether any type of lying is justified remains an issue of conflict for most of us, even though there are times when it seems justified.
This conflict over lying has become more pronounced over the last 50 years as the rules of etiquette, which in the past have been used to control much of social behavior, have been replaced by the idea that we should speak our minds—that is, not to lie by words or emotional behavior.
The change from fixed rules, independent of internal feeling, to frank expression of our feelings has intensified our ideas about lying and deception, making such behavior socially as well as morally unacceptable. This may explain why parents are so upset about their children’s lying.
When Margaret comes home from a play date with Rana she is holding a doll, which she claims was given to her by Rana’s mother. However, a phone call from Rana’s mother, mentioning that the doll can be brought back at the children’s next play date, reveals that Margaret has taken something that was not hers. Margaret’s false statement now becomes the central issue, as her mother says, “You lied to me! Now you will be punished.”
Thus, the focus of the problem has shifted: The point at issue is now the failure in the relationship between Margaret and her mother because Margaret has lied to her, rather than the girl’s moral failure in taking her friend’s doll. Had her mother known that lying to protect oneself from punishment is likely a natural and adaptive response of our species, she might not have lost the opportunity to educate Margaret on the fundamental moral issue in this incident, namely that of stealing.
The research up to now suggests strongly that lying is a human behavior and that most forms of deception have adaptive significance. It is fair to say, however, that our conflicted feelings about lying and deception probably reflect an inherent conflict between two evolutionarily derived needs: the need for some form of trust and for social harmony. In fact, Lee, when examining lies to protect others’ feelings and lying for protection, found that school-age children can make this distinction.
What we need to learn from the work carried out so far is that some forms of lying, for example, to protect ourselves from punishment, are necessary only because we have violated a social or moral sanction. It is toward this, the teaching of social and moral standards, that parents can most effectively direct their efforts for the sake of their children’s future well-being—not toward the eradication of lying, because the studies discussed here show that lying (whether to avoid punishment, to protect the feelings of others, or as a form of self-deception) may well constitute an important aspect of prosocial behavior.
Future research on the relation between lying and deception as they relate to subsequent moral behavior will be important to explore. We will need to continue to study how lying and deception affect social-emotional behavior and the forming and maintaining of adult social relationships. In addition, the relation between self-deception and psychopathology needs careful study, because the role of self-deception in the maintenance of self-esteem has important implications for the treatment of psychological distress as a consequence of traumas related to conflict and war.
Click "American Scientist" to access home page
American Scientist Comments and Discussion
To discuss our articles or comment on them, please share them and tag American Scientist on social media platforms. Here are links to our profiles on Twitter, Facebook, and LinkedIn.
If we re-share your post, we will moderate comments/discussion following our comments policy.