A Short History of Hydrogen Sulfide
By Roger P. Smith
From the sewers of Paris to physiological messenger
From the sewers of Paris to physiological messenger

DOI: 10.1511/2010.82.6
Early last year, reports began to emerge in the Southeastern United States of a strange illness. Homeowners reported nosebleeds, sinus irritation and respiratory problems that appeared to be associated with corrosion of copper pipes and air conditioner coils in their houses.
The culprit seems to be drywall imported from China and possibly contaminated with strontium sulfide, an unstable salt that releases hydrogen sulfide on exposure to moisture. It was used widely in the housing boom of 2004–2007, and in the rebuilding efforts after Hurricane Katrina in 2005, when domestic suppliers could not keep up with the demand. The Consumer Products Safety Commission is now investigating whether sulfide gases given off by the drywall, including hydrogen sulfide, are to blame. The Florida Department of Health maintains a Web site with information for consumers. Lawsuits abound, and many who are able to do so have moved out of their homes. Several estimates place the number of affected houses at 100,000.

Photographs courtesy of the Florida Department of Health.
To those experiencing or investigating this phenomenon early on, it seemed bizarre. But in fact, this is just the latest chapter in the history of a chemical whose effects were first noted in the 16th century. And there is still more to learn about its role in the human body. Recent research offers insights into its biochemical actions as well as some intriguing suggestions for medical uses.
In 1713, a remarkable Italian physician named Bernardino Ramazzini published De Morbis Artificum, or Diseases of Workers. In Chapter 14, titled “Diseases of Cleaners of Privies and Cesspits,” he describes a painful inflammation of the eyes which was common among such workers. The inflammation often led to secondary bacterial invasion, and sometimes to total blindness. Displaying amazing insight, Ramazzini hypothesized that when the cleaners disturbed the excrement in the course of their work, an unknown volatile acid was produced, which was irritating to the eyes. It was also at least partially responsible for the odor of excrement, and it is now known to be generated wherever organic matter undergoes putrefaction.
Ramazzini further postulated that that same acid was causing copper and silver coins which the workers had in their pockets to turn black on their surfaces—an eerie resonance with the phenomena recently observed by U.S. homeowners. Around 1777, a series of accidents—some of them fatal—began to occur in Paris due to a gas emanating from its sewer system. The commission appointed to study the cases made its findings public in 1785. The report described two distinct types of poisonings: a mild form involving inflammation of the eyes and mucous membranes as already described by Ramazzini, and a severe form that was characterized by a fulminating (rapidly developing) asphyxia. It is little wonder that the French Romantic writer Victor Hugo (1802–1885) referred to the Parisian sewers as “the intestine of the Leviathan.” Many years were to pass, however, before chemical analyses would establish the presence of hydrogen sulfide in the sewers and implicate it as the cause of the accidents.
The history of exposures has focused on sewers and the workplace, but the corrosive effects of hydrogen sulfide are common knowledge in Rotorua, New Zealand, which was built over centuries in a geothermally active area. The constant exposure to low, environmental levels of hydrogen sulfide produces such damage even as residents enjoy spas, indoor heating and cooking with the hot gases.
Today, the American Conference of Industrial Hygienists has set the so-called threshold limit value for its presence in the workplace at 10 parts per million (ppm) of hydrogen sulfide in air for eight hours a day, five days a week over a working lifetime. The U.S. National Institute for Occupational Safety and Health estimated in 1977—some 200 years after the Paris accidents—that 125,000 workers in at least 77 occupations, including drilling for petroleum, tanneries and the paper industry, may be at risk of exposure to hydrogen sulfide.
Around 1750, a humble young Swede beginning his career as an apothecary was fortunate to have a series of very understanding mentors who allowed him considerable free time for reading and experimentation. His name was Carl Wilhelm Scheele, and he turned out to be a gifted chemist. Like many chemists before and after him, Scheele seems to have given little thought to the biological effects of the materials with which he worked. One day while distilling potassium ferrocyanide with sulfuric acid, he noted a “strong, peculiar and not unpleasant odor.” He brought himself to taste this gas and described it as “slightly on sweet [sic] and somewhat heating on the mouth.” Today we describe the odor as that of bitter almonds and call the gas hydrogen cyanide. Scheele may have been fortunate to have escaped with his life.
Perhaps a guardian angel was with him again on the day that he treated ferrous sulfide (pyrite, or fool’s gold) with a mineral acid. He called the rank odor that resulted Schwefelluft (sulfur air) and referred to it as stinkende (stinking or fetid). Today we refer to the odor as that of rotten eggs. His patron, the Swedish chemist and mineralogist Torbern Olof Bergman, also demonstrated its presence in some mineral springs. The publication date for these original observations was 1777—around the time of the Paris accidents. The fact that the same man discovered both hydrogen sulfide and hydrogen cyanide was the start of a long series of coincidences and discoveries about the two chemicals that would uncover their similarities.
Investigations on the biological effects of hydrogen sulfide began around the turn of the 20th century. François Chaussier, François Magendie, Claude Bernard and Felix Hoppe-Seyler were among the well-known scientists of the day who labored in that vineyard.
In experiments with dogs, marked differences in the effects of hydrogen sulfide were noted with only small changes in its concentration in the air they were breathing. Fifty ppm, which was considered a minimally lethal concentration at the time, resulted in a slight progressive depression in the rate and depth of respirations. After many hours of exposure, the dogs died from a type of pulmonary edema, acute respiratory distress syndrome (ARDS). When that concentration was doubled to 100 ppm, death resulted in 15 to 20 minutes. In these cases the respiration was stimulated almost immediately, this progressed to a pronounced hyperpnea (deep breathing), and death in apnea followed. At 300 ppm, respiratory arrest occurred after a few violent gasps. The same effects on respiration, with the exception of ARDS, were known to occur with injected or inhaled hydrogen cyanide. Mice may be more resistant to the effects of hydrogen sulfide. In a 1964 experiment, they survived for 10 minutes in an atmosphere of 1,872 ppm and for 20 minutes at 985 ppm.
Adapted by Tom Dunne.
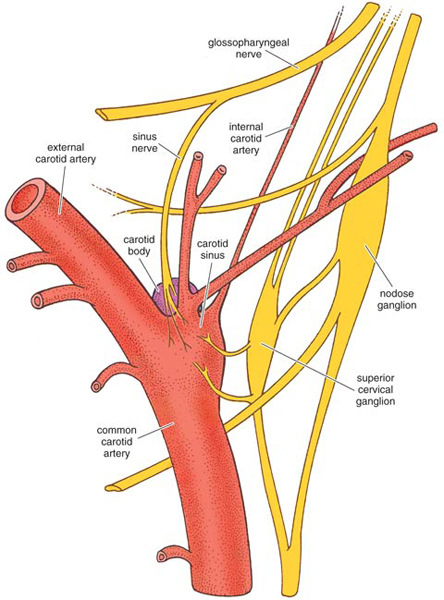
A more complete explanation of the respiratory stimulant effects of hydrogen cyanide and hydrogen sulfide had to await the discovery of the chemoreceptor function of the carotid body (shown at right), and the reflex effects that follow the activation of those receptors. The respiratory stimulant effects of cyanide and sulfide could be completely abolished by severing the sinus nerve and thus denervating the carotid sinus. In that case, larger doses of either chemical resulted only in respiratory depression, which was presumably mediated via the brainstem. Similarly, injections of sulfide or cyanide into the internal carotid or vertebral arteries also failed to stimulate respiration, since in those cases the chemicals reached the carotid sinus only after dilution in the general circulation. When innervation of the sinus was intact, the hyperpnea was accompanied by a fleeting rise in systemic blood pressure, and sometimes by a slowed heart rate (bradycardia). The cardiovascular effects varied with the injection site and the species and are still not adequately explained.
The great German biochemist Hoppe-Seyler became famous for his discovery of the abnormal form of hemoglobin known as methemoglobin, in which some or all of the heme irons have been oxidized to the ferric form. This reaction is readily mediated both in vivo and in vitro by sodium nitrite. Methemoglobin cannot reversibly combine with oxygen, and the disruption of the oxygen-transport function of the blood can result in hypoxia and death.
In 1863, Hoppe-Seyler passed a stream of pure hydrogen sulfide through a sample of human blood and claimed to have observed a greenish pigment that was associated with shifts in the visible absorption spectrum of hemoglobin. Although he was aware that he had probably produced a mixture containing unstable and denatured products, which resulted in turbidity and precipitation and made the absorption spectra suspect, he still thought that the mixture contained a new form of hemoglobin. He called it sulfhemoglobin and thereby launched one of the most confused areas in hematology. It led to the hypothesis some still subscribe to—namely, that hydrogen sulfide is a blood poison like sodium nitrite and carbon monoxide. No matter that animal experiments clearly demonstrated that it was a respiratory toxin, or that no such pigment has ever been identified in the blood of animals or humans fatally poisoned with hydrogen sulfide. Sulfhemoglobin generated by hydrogen sulfide appears to be a strictly in vitro phenomenon, and it has yet to be prepared in pure form.
No less a scientist than Linus Pauling and his associates described the magnetic properties of another blood pigment, which they called sulfmethemoglobin. This pigment is easily prepared in pure form by mixing hydrogen sulfide with methemoglobin, and it is chemically analogous to cyanmethemoglobin, in which the cyanide ion is bound to ferric irons of heme. The hydrosulfide anion (HS–) also binds to ferric heme, albeit not quite so tenaciously as cyanide. Indeed, this reaction has been exploited medically as an antidote to cyanide poisoning. One can deliberately inject sodium nitrite intravenously to generate a tolerable level of methemoglobin. The methemoglobin will temporarily bind free cyanide as the inactive complex cyanmethemoglobin. Over time the cyanide is slowly released, at a rate at which the body’s natural detoxification mechanisms can deal with it.
At least three laboratories have demonstrated that the same principles can be applied to hydrogen sulfide poisoning and that induced methemoglobinemia can indeed be lifesaving. At least a half dozen successful human resuscitations have been reported in the literature. The odds against its successful application, however, are high. Few poisons are more rapidly acting than inhaled hydrogen sulfide, and inhalation is invariably the route of exposure. Sulfide poisoning tends to occur in remote locations, and there is seldom a medically qualified individual on the scene who is prepared with a parenteral form of nitrite and trained to make intravenous injections. Most successful resuscitations from cyanide poisoning have occurred in individuals who ingested soluble salts of cyanide, where absorption is delayed.
In addition to the similarities in their physiological effects, cyanide and sulfide have chemical similarities. The undissociated forms of both hydrogen cyanide and hydrogen sulfide are flammable, volatile gases. Hydrogen sulfide (whose vapor density, or d, is 1.19) is heavier than air (d = 1.0) whereas hydrogen cyanide (d = 0.941) is lighter. Both are weak acids with acid dissociation constants (pKa) that are of some physiological significance: hydrogen cyanide 9.2–9.3 and hydrogen sulfide 7.04. Both form salts with sodium and potassium as well as with some alkaline earths. Both anions bind to methemoglobin as noted above, and each of those complexes has its distinct visible absorption spectrum. And both are inhibitors of cytochrome c oxidase, the terminal enzyme in the electron transport chain that reacts with molecular oxygen in aerobic organisms. Blockage of that key enzyme is believed to be the mechanism of action in the rapidly lethal form of cyanide and sulfide poisonings.
Inhibition of cytochrome c oxidase results in a decrease in oxidative phosphorylation (the metabolic pathway that produces ATP). This in turn lowers the metabolic rate and body temperature in mice. These phenomena accompany states of suspended animation. When mice were exposed to hydrogen sulfide at concentrations of 80 ppm, dramatic effects were observed in the first 5 minutes. By 6 hours, their metabolic rate had dropped by 90 percent and body temperature to 2 degrees Celsius above ambient temperature. When the mice were then returned to room air, the metabolic rate and temperature returned to normal with no detectable behavioral or functional deficits. As already noted, lethal levels for hydrogen sulfide in mice are much higher than for dogs, but this state in mice must also occur over a fairly narrow range of concentrations.
Finally, and most astonishingly of all, experimental evidence contributed in 2008 indicates that like carbon monoxide and nitric oxide, hydrogen sulfide is an important signaling molecule in biology, and it may find a role in medicine. It is physiologically generated in mice by cystathionine γ-lyase, and genetic deletion of that enzyme markedly reduces hydrogen sulfide levels in the serum, heart, aorta and other tissues. Mutant mice lacking the enzyme have marked hypertension and diminished endothelium-dependent vasorelaxation, consistent with an important vasodilator role for hydrogen sulfide. The enzyme is physiologically activated by the calcium-binding protein calmodulin, which is a mechanism for hydrogen sulfide formation in response to vascular activation. Thus, hydrogen sulfide appears to be a physiologic vasodilator and regulator of blood pressure. Its relative contribution vis-à-vis the similar nitric oxide is not yet clear.
What a strange and wondrous journey this odiferous and violently toxic chemical, associated with the excrement of humanity, has led us on for five centuries. It’s a history that could fill a book, one that covers a vast range of territory, from the search to determine the cause of workplace injuries to fascinating discoveries about how hydrogen sulfide interacts with chemoreceptors in the body. And for all the false leads, in the end it may yet turn out to have some useful applications in medicine—even if only a new Viagra.
Click "American Scientist" to access home page
American Scientist Comments and Discussion
To discuss our articles or comment on them, please share them and tag American Scientist on social media platforms. Here are links to our profiles on Twitter, Facebook, and LinkedIn.
If we re-share your post, we will moderate comments/discussion following our comments policy.